Pooling data for primary total knee implants across national registries: is the same implant used in multiple registries and for the same patient group? An observational study
Lotje A HOOGERVORST 1, Rob G H H NELISSEN 1, Liza VAN STEENBERGEN 2, Alma B PEDERSEN 3,4, Eskild Bendix KRISTIANSEN 3, Martin LINDBERG-LARSEN 5,6, Marina TORRE 7, Enrico CIMINELLO 7, Riccardo VALENTINI 7, Alexander W GRIMBERG 8, Yinan WU 8, and Perla J MARANG-VAN DE MHEEN 1,9
1 Department of Orthopaedics, Leiden University Medical Center, Leiden, the Netherlands; 2 Dutch Arthroplasty Register (LROI), ‘s- Hertogenbosch, the Netherlands; 3 Department of Clinical Epidemiology, Aarhus University Hospital, Aarhus, Denmark; 4 Department of Clinical Medicine, Aarhus University, Aarhus, Denmark; 5 The Danish Knee Arthroplasty Register; 6 Department of Orthopedic Surgery and Traumatology, Odense University Hospital, Odense, Denmark; 7 Italian Implantable Prostheses Registry, Italian National Institute of Health, Rome, Italy; 8 German Arthroplasty Registry – EPRD, Berlin, Germany; 9 Safety & Security Science and Centre for Safety in Healthcare, Delft University of Technology, Delft, the Netherlands
Background and purpose — Pooling data on the performance of total knee (TK) implants across registries is only possible if the same TK implant is used across multiple registries and if used in patients with similar characteristics. We assessed to what extent specific TK implants: (i) are used across multiple registries or only in a single registry; and (ii) differ in patient characteristics between registries.
Methods — All primary TK implants implanted between January 2020 and December 2021 in the Danish, Dutch, German, and Italian registries were included. We determined the number of registries using a specific TK implant (based on combined femoral-tibial component brand name and fixation/congruency/mobile bearing insert/patella usage). Patient characteristics (age/body mass index [BMI]/sex/diagnosis osteoarthritis) were compared across registries for TK implants used in ≥ 2 registries ≥ 100 times.
Results — 813 different TK implants (577,351 procedures) were used across the 4 registries, of which 53 TK implants (7%) were used in 1 registry (8,000 procedures). 760 different TK implants (569,351 procedures; 99%) were used in ≥ 2 registries of which 47 different TK implants (393,954 procedures; 68%) were used in ≥ 2 registries and ≥ 100 times. Statistically and clinically significant differences in age for the same TK implant across registries were observed for 29 TK implants (62%) and 3 TK implants (6%), respectively; for other characteristics these were for BMI 30 (64%) and 0 (0%) TK implants; for male proportion 23 (49%) and 17 (36%) TK implants; and for diagnosis of osteoarthritis 42 (89%) and 34 (72%) TK implants, respectively.
Conclusion — Most specific TK implants and TK procedures were used across multiple registries, but they were often used in patients with different characteristics. This has an impact on comparing implant performances between registries.
Citation: Acta Orthopaedica 2025; 96: 339–347. DOI: https://doi.org/10.2340/17453674.2025.43476.
Copyright: © 2025 The Author(s). Published by MJS Publishing – Medical Journals Sweden, on behalf of the Nordic Orthopedic Federation. This is an Open Access article distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/)
Submitted: 2024-12-05. Accepted: 2025-03-23. Published: 2025-04-17.
Correspondence: l.a.hoogervorst@lumc.nl
LAH: conceptualization, data curation, formal analysis, methodology, project administration, writing—original draft. RGHHN: conceptualization, funding acquisition, writing—review and editing. PJM-vdM: conceptualization, funding acquisition, data curation, methodology, project administration, writing—review and editing. ABP, AWG, EBK, MLL, EC, LvS, MT, RV, YW: data curation and writing—review and editing.
The authors would like to thank Paola Laricchiuta and Filippo Boniforti for their contribution to the Italian Arthroplasty Registry and the Danish, Dutch, German, and Italian arthroplasty registries for providing the data, as well as all the operating theatre staff at participating hospitals throughout these countries for collecting the data.
Handling co-editors: Marianne Westbergand Robin Christensen
Acta thanks Oystein Johannes Gothesen and Peter Lawrence Lewis for help with peer review of this manuscript.
Arthroplasty registries are well suited to assess the safety and performance of total knee (TK) implants, as most registries publish annual reports including survivorship data of specific TK implants [1-3]. Many registries have outlier procedures in place to detect implants with significantly higher revision [1,2,4]. Several factors may influence TK-implant performances, including implant-related factors such as implant materials or the production process, for which the Optetrak case showed that implant-related factors resulted in significantly worse performance [5-7].
Patient characteristics (e.g., age, sex, and body mass index [BMI]) can also affect the performance of TK implants [8-10]. To compare performance of a specific a TK implant across registries, characterized by brand name and implant characteristics to avoid camouflage, it is thus important to consider the characteristics of patients receiving that specific TK implant. Few studies have assessed differences in patient characteristics across countries [11-13], with most studies only focusing on variations in preoperative pain and function. Importantly, all studies analyzed the entire group of TK implants (e.g., all cemented TK implants) rather than analyzing differences for specific TK implants (characterized by a specific brand and implant characteristics like fixation and congruency). Hence, a more comprehensive analysis of the similarities and differences in patients receiving a specific TK implant is required to better understand possible differences in safety and performance of TK implants across registries. Such a comprehensive analysis is also needed to pool data across countries/registries or when performing distributed meta-analyses, where ensuring the same patient mix is crucial for fair comparison of safety and performance.
The aim of this study was to assess, across national registries, to what extent specific TK implants: (i) are used across multiple registries or only in a single registry; and (ii) differ in patient characteristics between registries.
Methods
Design and setting
The study was designed as a comparative observational study including data from 4 national European arthroplasty registries. 8 European national registries were asked to participate. Although all registries showed interest in doing so, this required some effort to make the standard script applicable to the registry, conduct the analyses, and send the data, which the following 4 European registries managed to do: the Danish Knee Arthroplasty Register (DKR), the Dutch Arthroplasty Register (LROI), the German Arthroplasty Register (EPRD), and the Italian Arthroplasty Registry (RIAP). Regarding the EPRD, only registry data with complete linkage to insurance data was included (10.5% of all TK procedures) [14]. TK implant-level completeness for the included 4 registries ranged from 59% (RIAP) to 97% (LROI) [15,16]. Aggregated TK-implant-level data was retrieved from each registry, including all patients receiving a primary TK implant between January 1, 2010 and December 31, 2021. In addition, each registry provided the number of unicompartmental knee (UK) implant procedures in this period, as different use of UK implants across registries may have been impacted by patient characteristics [13]. The study is reported according to STROBE guidelines.
Categorization of TK implants
Groups of comparable TK-implant constructs were defined based on the following implant characteristics: implant–bone fixation (i.e., fixation), tibial insert–femoral congruency (i.e., congruency), mobile bearing insert, and patella usage (Table 1). Within each TK construct, the brand name of both the femoral and tibial component was used to indicate a specific TK implant.
| Fixation: | Cemented; cementless; hybrid – LROI definition: femoral component is cementless, tibial and/or patellar components are cemented |
| Congruency: | Mega prosthesis (i.e., maximal-hinged or mega tumor resection prosthesis) – LROI definition of hinged: a component that only allows for flexion and extension through a fixed axis and provides collateral as well as posterior ligament stability |
| Fully congruent (high posterior peg of liner) – LROI definition of fully: a component that only allows for flexion and extension through a fixed axis and provides collateral as well as posterior ligament stability | |
| Posterior (i.e., posterior stabilized) – LROI definition: both cruciate ligaments removed | |
| Medial pivot – LROI definition: the medial pivot knee design has a highly congruent medial liner–femoral component contact | |
| Minimal (i.e., minimally congruent): retaining of posterior cruciate ligament (CR) – LROI definition: retaining of medial CR | |
| Bicruciate retaining – LROI definition: both cruciate ligaments retained | |
| Bearing insert: | Fixed (i.e., non-mobile) – LROI definition: component that is not intended to move relative to its interface component |
| Mobile/rotating (i.e., a tibial insert is intended to move on its metal tibial component) | |
| LROI definition of mobile: a component that is intended to move relative to its interface component | |
| LROI definition of rotating: a component that is intended to move relative to its interface component. Rotating: where the component moves in an inward and outward direction | |
| Patella usage: | No; yes |
Patient characteristics
For each specific TK implant in a registry, the number of procedures as well as the following patient characteristics were retrieved: (i) mean age (standard deviation [SD]); (ii) mean BMI (SD); (iii) percentage male sex, and (iv) percentage of patients with the diagnosis osteoarthritis. The registers differed in their classification of the initial diagnosis, and we calculated the percentage of patients with the diagnosis osteoarthritis in the following way for each registry:
- DKR: the number of patients with primary osteoarthritis as the initial diagnosis, relative to the total number of patients. Other diagnoses include rheumatoid arthritis, sequelae after tibia/femur condyle fracture, sequelae after patellar fracture, secondary arthrosis after meniscectomy, hemophilia, cancer, or other.
- EPRD: the number of patients with primary osteoarthritis as the initial diagnosis, relative to the total number of patients. Other diagnoses include post-traumatic osteoarthritis, secondary osteoarthritis, or other.
- LROI: the number of patients with osteoarthritis as the initial diagnosis, relative to the total number of patients. Other diagnoses include post-traumatic, rheumatoid arthritis, osteonecrosis, or other.
- RIAP: the number of patients with primary osteoarthritis as the initial diagnosis, relative to the total number of patients. Other diagnoses include post-traumatic osteoarthritis, rheumatoid arthritis, neoplasia, osteonecrosis, or other.
Statistics
Descriptive statistics were used to assess the number of registries in which each TK construct (based on implant characteristics: fixation, congruency, mobile bearing insert, and patella usage) was used. We also calculated the percentage of UK-implant procedures reported in each registry relative to all knee (i.e., both TK and UK) implant procedures used.
For each specific TK implant used in ≥ 2 registries and used ≥ 100 times in each registry, we compared patient characteristics across registries. The criterion of ≥ 100 TK implants used per registry was added to ensure sufficient sample size for meaningful analysis. First, we calculated for all patients receiving a TK implant across registries: the mean (SD) age and BMI, as well as the percentage of male sex and patients with osteoarthritis. Thereafter, for each registry and specific TK implant, we calculated a confidence interval around the mean or percentage, using the SD and total number of patient procedures. Statistically significant differences were determined by non-overlapping confidence intervals between registries [17]. As statistical significance does not equal clinical relevance, we applied the commonly used threshold of a ≥ 10% difference (i.e., 10% difference on the 0 to 100% percentage scale) to determine a clinically relevant difference for male sex and osteoarthritis diagnosis, and for the continuous variables age and BMI we used thresholds of a ≥ 5 years difference and a ≥ 5 points difference, respectively [18,19]. These commonly used thresholds are determined in a large cohort study (including 4,183 patients) and in a Delphi study (i.e., the assessment of quality in the lower limb Arthroplasty “AQUILA” initiative) including 44 orthopedic experts.
Ethics, data sharing plan, funding, use of AI, and disclosures
This work was supported by the European Union Horizon 2020 Research and Innovation Program (grant number 965246) and was part of the Coordinating Research and Evidence for Medical Devices (CORE-MD) project. AI tools were not used in our submission. Complete disclosure of interest forms according to ICMJE are available on the article page, doi: 10.2340/17453674.2025.43476
Results
Inclusion of TK constructs
Based on the combination of implant characteristics (i.e., 3 fixation types, 6 congruency types, 2 mobile bearing insert types [yes/no], and patella usage [yes/no]) 72 TK constructs would be possible theoretically, of which 9 (13%) were not used in any of the 4 registries or did not exist (Figure). 63 TK constructs (577,351 procedures; 813 different TK implants) were used in the 4 registries. Of these, 25 (40%) TK constructs were used in a single registry, including 53 out of 813 (7%) different TK implants and 8,000 out of 577,351 (1%) procedures. 27 (71%) of the remaining 38 TK constructs (175,397 procedures; 713 different TK implants) did not have specific TK implants used ≥ 100 times in ≥ 2 registries. Thus, 11 TK constructs, considering 47 specific TK implants and 393,954 (68%) procedures, were included in the comparison of patient characteristics between registries (Figure, Table 2). Of note, no specific TK implants with mobile/rotating bearing inserts were used ≥ 100 times in ≥ 2 registries. Overall, 206 TK implants with mobile/rotating bearing inserts were used across registries but 183 of these were used in only 1 registry and 23 TK implants with mobile/rotating bearing inserts were used < 100 times.
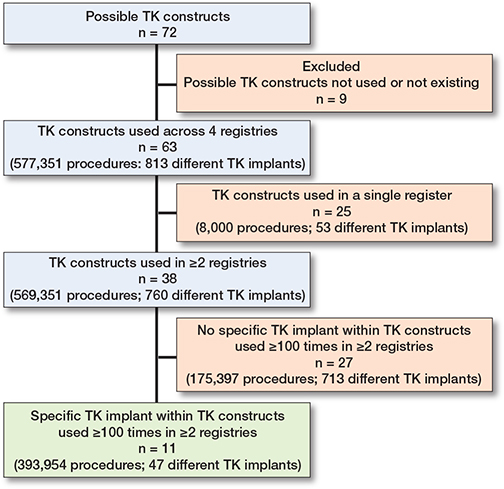
Use of specific TK implants across registries and included TK implants in comparison of patient characteristics.
Inclusion of TK implants
Table 2 shows the femoral–tibial brand name combinations for the 47 specific TK implants included in each of the 11 TK constructs, as well as the number of procedures in which these TK implants were used. Most included TK-implant procedures were registered in the LROI (n = 207,017) followed by the EPRD (n = 139,940), the DKR (n = 41,526), and the RIAP (n = 5,471). In 4 TK constructs, only 1 specific TK implant was used, while the TK construct “cemented, minimal congruent, fixed, and no patella usage” included the highest number of specific TK implants, namely 13. 3 TK constructs were used in all 4 registries: (i) the cemented, minimal congruent, fixed, without patella (including 13 different TK implants); (ii) the cemented, posterior stabilized, fixed, with patella (including 7 different TK implants), and (iii) the hybrid, minimal congruent, fixed, without patella (including 5 different TK implants). 5 TK constructs were used in 3 registries, and 3 TK constructs were used in 2 registries.
Use of UK implants across registries
The percentage of UK implants used was rather similar across registries: highest in the DKR (15.3%), followed by the LROI (13.3%), RIAP (12.4%), and the EPRD (12.1%).
Comparing patient characteristics for specific TK implants between registries
Overall, patients receiving the 47 specific TK implants were on average 68 years old (SD 7.3 years), with a mean BMI of 30 (SD 3.4), 34% were male, and 81% had the diagnosis osteoarthritis.
Of these 47 TK implants, statistically significant differences in age between registries were found for 29 (62%) TK implants, 30 (63%) had differences in BMI, 23 (49%) in percentages of male sex, and 42 (89%) in percentage of patients with osteoarthritis (Tables 3 and 4). Only 1 TK implant, Genesis II–Genesis II (hybrid/fixed/posterior stabilized/with patella) had no statistically significant difference between registries for any of these patient characteristics.
| TK construct and TK implant | n | Mean age | Mean BMI | ||||||
| DKR | EPRD | LROI | RIAP | DKR | EPRD | LROI | RIAP | ||
| C/F/F/No patella | |||||||||
| NexGen–NexGen | 991 | – | 72 | 72 | – | – | 31 | 29 a | – |
| C/MP/F/No patella | |||||||||
| Evolution MP–Evolution MP | 2,561 | – | 68 | 69 | 72 b | – | 31 | 29 a | – |
| Advance–Advance | 546 | – | 71 | 71 | – | – | 31 | – | – |
| C/MP/F/Patella | |||||||||
| Evolution MP–Evolution MP | 399 | – | 65 | 68 | – | – | 30 | 29 | – |
| C/M/F/No patella | |||||||||
| Vanguard Complete–Vanguard Complete | 37,811 | 68 | 67 b | 70 | – | 29 | 29 b | 31 | – |
| PFC/Sigma–PFC/Sigma | 31,435 | 68 | 70 b | 69 | – | 29 | 31 b | 30 | – |
| NexGen–NexGen | 21,740 | 68 | 70 b | 68 | – | 29 | 31 b | 29 | – |
| Genesis II–Genesis II | 18,731 | – | 68 | 69 a | – | – | 31 | 30 a | – |
| Columbus–Columbus | 13,757 | – | 69 | – | 72 | – | 31 | – | – |
| Triathlon–Triathlon | 11,672 | 67 d | 69 | 69 | 72 c,d | 30 | 31 b | 30 | – |
| Persona–Persona | 7,517 | – | 69 | 65 d | 71 b,d | – | 31 a | 28 | – |
| TC–plus–TC–plus | 5,832 | – | 71 a | 69 | – | – | 31 | 31 | – |
| Attune–Attune | 5,678 | – | 66 d | 72 a,d | – | – | 31 | 29 a | – |
| ACS–ACS | 3,875 | – | 67 | 66 | – | – | 32 | 29 a | – |
| Balansys–Balansys | 3,754 | – | 70 a | 68 | – | – | 31 | 31 | – |
| Innex–Innex | 2,305 | – | 71 | 74 a | – | – | 31 | – | – |
| Unity–Unity | 427 | – | 70 | – | 71 | – | 31 | – | – |
| C/M/F/Patella | |||||||||
| PFC/Sigma–PFC/Sigma | 21,468 | 70 | 70 | 69 b | – | 30 | 31 b | 30 | – |
| Vanguard Complete–Vanguard Complete | 10,704 | 67 b | 70 | 68 | – | 30 | 31 b | 30 | – |
| NexGen–NexGen | 5,766 | 69 a | 71 | – | – | 30 | 31 a | – | – |
| Triathlon–Triathlon | 5,099 | 67 a | 68 | 68 | – | 30 | 31 | 30 | – |
| Genesis II–Genesis II | 3,056 | – | 67 | 68 | – | – | 32 a | 30 | – |
| C/P/F/No patella | |||||||||
| NexGen–NexGen | 55,367 | – | 69 | 69 a | – | – | 31 | 29 a | – |
| Genesis II–Genesis II | 37,974 | – | 69 | 69 | – | – | 31 | 30 a | – |
| Triathlon–Triathlon | 7,665 | – | 70 | 70 | – | – | 31 | 30 a | – |
| Persona–Persona | 6,648 | – | 70 | 68 a | 71 | – | 30 | 29 a | – |
| PFC/Sigma–PFC/Sigma | 5,845 | – | 70 | 69 a | – | – | 31 | 30 a | – |
| Balansys–Balansys | 3,809 | – | 70 | 69 a | – | – | 31 | 29 a | – |
| Attune–Attune | 2,726 | – | 69 | 69 | 72 b | – | 31 | 30 | – |
| ACS–ACS | 601 | – | 69 | 66 a | – | – | 32 | 31 | – |
| Columbus–Columbus | 536 | – | 68 | – | 72 a | – | 32 | – | – |
| C/P/F/Patella | |||||||||
| NexGen–NexGen | 14,523 | 68 | 69 a | 68 | – | 30 | 32 b | 29 | – |
| Genesis II–Genesis II | 12,204 | – | 69 | 67 a | – | – | 30 | 30 | – |
| PFC/Sigma–PFC/Sigma | 8,729 | 68 a | 69 | 69 | – | 30 | 32 b | 30 | – |
| Triathlon–Triathlon | 1,980 | – | 70 | 68 a | – | – | 31 a | 30 | – |
| Balansys–Balansys | 1,462 | – | 69 | 66 a | – | – | 32 | 29 a | – |
| Attune–Attune | 1,273 | – | 69 | 69 | 71 b | – | 31 | 30 | – |
| Persona–Persona | 794 | – | 68 | 66 a | 70 | – | 31 | 29 a | – |
| H/M/F/No patella | |||||||||
| Vanguard Complete–Vanguard Complete | 3,134 | 67 | 68 | 69 | – | 30 | 31 | 29 a | – |
| NexGen–NexGen | 3,074 | 69 | 68 a | 69 | – | 29 b | 31 | 31 | – |
| PFC/Sigma–PFC/Sigma | 807 | 69 | 68 | – | – | 29 a | 31 | – | – |
| Triathlon–Triathlon | 430 | 70 | 69 | – | – | 30 | 30 | – | – |
| Columbus–Columbus | 478 | – | 70 | – | 70 | – | 31 | – | – |
| H/M/F/Patella | |||||||||
| NexGen–NexGen | 4,984 | 68 | 68 | – | – | 30 a | 31 | – | – |
| Vanguard Complete–Vanguard Complete | 624 | 67 | – | 69 | – | 30 | – | 30 | – |
| H/P/F/Patella | |||||||||
| Genesis II–Genesis II | 333 | – | 69 | 66 | – | – | 30 | 31 | – |
| U/M/F/No patella | |||||||||
| Triathlon–Triathlon | 2,830 | 67 | 66 a | 68 | – | 30 | 31 | 29 b | – |
| a Statistically significantly difference from 1 other registry; b Statistically significantly different from 2 other registries; c Statistically significantly different from 3 other registries; d Clinically relevant difference. |
|||||||||
| TK construct and TK implant | n | Proportion male (%) | Proportion osteoarthritis (%) | ||||||
| DKR | EPRD | LROI | RIAP | DKR | EPRD | LROI | RIAP | ||
| C/F/F/No patella | |||||||||
| NexGen–NexGen | 991 | – | 28 | 22 | – | – | 83 a,d | 100 d | – |
| C/MP/F/No patella | |||||||||
| Evolution MP–Evolution MP | 2,561 | – | 34 | 34 | 29 | – | 93 b | 100 | 99 |
| Advance–Advance | 546 | – | 41 d | 10 a,d | – | – | 89 a,d | 100 d | – |
| C/MP/F/Patella | |||||||||
| Evolution MP–Evolution MP | 399 | – | 19 a,d | 33 d | – | – | 89 a,d | 100 d | – |
| C/M/F/No patella | |||||||||
| Vanguard Complete–Vanguard Complete | 37,811 | 40 | 48 d | 33 b,d | – | 100 d | 78 b,d | 93 d | – |
| PFC/Sigma–PFC/Sigma | 31,435 | 47 b,d | 36 d | 34 d | – | 83 d | 85 d | 100 b,d | – |
| NexGen–NexGen | 21,740 | 44 | 37 b | 43 | – | 85 b,d | 94 | 100 d | – |
| Genesis II–Genesis II | 18,731 | – | 34 | 35 | – | – | 93 a | 100 | – |
| Columbus–Columbus | 13,757 | – | 33 | – | 32 | – | 87 a,d | – | 99 d |
| Triathlon–Triathlon | 11,672 | 45 | 38 d | 36 d | 53 c,d | 79 c,d | 82 d | 100 d | 98 d |
| Persona–Persona | 7,517 | – | 39 d | 56 b,d | 38 d | – | 93 b | 100 | 98 |
| TC–plus–TC–plus | 5,832 | – | 37 a | 30 | – | – | 80 a,d | 100 d | – |
| Attune–Attune | 5,678 | – | 41 | 38 | – | – | 89 a,d | 100 d | – |
| ACS–ACS | 3,875 | – | 29 a | 37 | – | – | 79 a,d | 100 d | – |
| Balansys–Balansys | 3,754 | – | 32 | 32 | – | – | 88 a,d | 100 d | – |
| Innex–Innex | 2,305 | – | 35 | 27 | – | – | 87– | – | |
| Unity–Unity | 427 | – | 29 d | – | 42 d | – | 79 a,d | – | 91 d |
| C/M/F/Patella | |||||||||
| PFC/Sigma–PFC/Sigma | 21,468 | 39 b | 34 | 30 | – | 86 d | 86 d | 100 b,d | – |
| Vanguard Complete–Vanguard Complete | 10,704 | 41 b,d | 26 d | 30 d | – | 81 d | 91 d | 100 d | – |
| NexGen–NexGen | 5,766 | 38 | 35 | – | – | 94 | 85 a | – | – |
| Triathlon–Triathlon | 5,099 | 38 d | 40 d | 25 b,d | – | 81 d | 77 d | 100 b,d | – |
| Genesis II–Genesis II | 3,056 | – | 30 | 29 | – | – | 93 | 100 | – |
| C/P/F/No patella | |||||||||
| NexGen–NexGen | 55,367 | – | 32 a | 36 | – | – | 89 a,d | 100 d | – |
| Genesis II–Genesis II | 37,974 | – | 35 | 37 | – | – | 89 a,d | 100 d | – |
| Triathlon–Triathlon | 7,665 | – | 36 | 36 | – | – | 79 a,d | 100 d | – |
| Persona–Persona | 6,648 | – | 39 | 41 | 38 | – | 78 d | 100 d | 96 d |
| PFC/Sigma–PFC/Sigma | 5,845 | – | 33 a | 41 | – | – | 88 a,d | 100 d | – |
| Balansys–Balansys | 3,809 | – | 43 | 36 a | – | – | 96 a | 100 | – |
| Attune–Attune | 2,726 | – | 38 | 46 b,d | 32 d | – | 89 b,d | 100 d | 98 d |
| ACS–ACS | 601 | – | 36 | 32 | – | – | 89 d | 0 d | – |
| Columbus–Columbus | 536 | – | 34 | – | 29 | – | 87 | – | 94 |
| C/P/F/Patella | |||||||||
| NexGen–NexGen | 14,523 | 29 | 30 | 31 | – | 77 b,d | 93 d | 100 d | – |
| Genesis II–Genesis II | 12,204 | – | 18 a,d | 31 d | – | – | 86 a,d | 100 d | – |
| PFC/Sigma–PFC/Sigma | 8,729 | 40 b | 32 | 35 | – | 77 d | 74 d | 100 b,d | – |
| Triathlon–Triathlon | 1,980 | – | 34 | 34 | – | – | 72 a,d | 100 d | – |
| Balansys–Balansys | 1,462 | – | 37 d | 21 a,d | – | – | 99 | 100 | – |
| Attune–Attune | 1,273 | – | 39 | 39 | 33 | – | 57 b,d | 100 d | 98 d |
| Persona–Persona | 794 | – | 39 d | 28 d | 29 d | – | 91 b | 100 | 98 |
| H/M/F/No patella | |||||||||
| Vanguard Complete–Vanguard Complete | 3,134 | 40 | 46 | 38 | – | 87 d | 92 | 100 b,d | – |
| NexGen–NexGen | 3,074 | 42 d | 50 d | 29 b,d | – | 92 d | 82 b,d | 100 d | – |
| PFC/Sigma–PFC/Sigma | 807 | 43 | 41 | – | – | 86 | 86 | – | – |
| Triathlon–Triathlon | 430 | 49 d | 38 d | – | – | 86 d | 70 a,d | – | – |
| Columbus–Columbus | 478 | – | 34 | – | 34 | – | 92 a | – | 100 |
| H/M/F/Patella | |||||||||
| NexGen–NexGen | 4,984 | 41 | 39 | – | – | 90 d | 74 a,d | – | – |
| Vanguard Complete–Vanguard Complete | 624 | 40 d | – | 25 a,d | – | 74 a,d | – | 100 d | – |
| H/P/F/Patella used | |||||||||
| Genesis II–Genesis II | 333 | – | 44 | 48 | – | – | 98 | 100 | – |
| U/M/F/No patella | |||||||||
| Triathlon–Triathlon | 2,830 | 43 | 47 d | 37 a,d | – | 87 d | 93 | 100 b,d | – |
| a Statistically significantly difference from 1 other registry; b Statistically significantly different from 2 other registries; c Statistically significantly different from 3 other registries; d Clinically relevant difference. |
|||||||||
As for clinically relevant differences in patient characteristics when the same TK implant was used, age was different in 3 of the 47 (6%) TK implants, percentage of male sex in 17 (36%), percentage of patients with diagnosis of osteoarthritis in 34 (72%), whilst no differences in BMI were found.
Discussion
This is the first multi-registry study to compare the use of specific TK implants across registries and by comparing their use in comparable patients characterized by age, sex, BMI, and diagnosis of osteoarthritis. This is essential for comparison of safety and performance of the implant between registries. Only 53 (7%) of the 813 specific TK implants were used in a single registry, suggesting that pooling data across registries to detect any safety concerns is possible for most (93%) TK implants. Of the 47 TK implants used ≥ 100 times in ≥ 2 registries, statistically significant differences in patient characteristics were found in 62% of the TK implants for age, 77% for BMI, 49% for male sex, and 89% for diagnosis of osteoarthritis. Only a small number of these statistically significant differences in age and male sex were deemed clinically relevant, none for BMI, but a large proportion (72%) of the differences in osteoarthritis diagnosis. These findings suggest that when comparing the performance for specific TK implants across registries potential differences in patient characteristics should be considered, particularly regarding diagnosis.
Most studies investigating differences in patient characteristics across registries did not consider specific TK implants but analyzed all TK implants combined [11,12], and found considerable differences between countries in preoperative patient characteristics (e.g., age and BMI) and pain levels. Our study contributes to this literature by providing a more comprehensive analysis of differences in patient characteristics for specific TK implants. For clinicians, such detailed analysis on the TK-implant level will likely be more clinically relevant in guiding implant selection, as clinicians select implants based on their performance but need these patient characteristics to put the performance in context. For example, if the revision risk of a specific implant is good but based on a relatively older population while the patient concerned is much younger, then it is uncertain whether the implant will perform similarly in that patient. Such TK-implant-level information is also relevant for regulators to better interpret the safety and performances of TK implants on the market across registries [20], as elderly patients, for example, may have lower remaining life expectancy, and surgeons may be less likely to revise given the higher risks associated with surgery [21].
Even though we found statistically significant differences in age, BMI, percentage of males, and osteoarthritis diagnosis across registries for many TK implants, the question is whether these differences are also clinically relevant. In a large study population, even very small differences can be detected as statistically significant, though they may not be clinically relevant [22]. As clinical differences are more relevant for clinicians, we also determined the clinically relevant differences by applying commonly used thresholds [18,19]. Only a small number of TK implants showed clinically relevant differences in age, BMI, and male sex, but differences in osteoarthritis diagnosis remained for a large proportion of TK implants. This suggests that most TK implants are used in similar patient groups except for diagnosis. This is in line with research showing differences in treatment approaches for knee osteoarthritis between countries, influenced by several factors such as variations in healthcare systems, guidelines and preferred approaches, economic factors, and cultural preferences [23,24]. The relatively high differences in osteoarthritis diagnosis might be caused by differences in definitions or the classification used. While the DKR, EPRD, and RIAP included primary osteoarthritis to calculate the percentage, the LROI included both primary and secondary osteoarthritis. Even though we tried to harmonize as much as possible across registries, these differences reflect the heterogeneity in definitions and methods across registries and show the need for further harmonization for better comparison.
To allow for early detection of safety issues in specific implants, it is often recommended that data across registries should be pooled to increase the number of implants at risk for statistical analysis and thereby statistical power [2,25]. Another advantage of pooling data across registries is that it might better represent real-world performance of this specific device across all patients in which it is used. On the other hand, if we want to know the revision risk for a specific implant in a specific patient population, we would need to include only specific patients to arrive at the best revision risk estimate, akin to what we do when pooling data in a meta-analysis. In addition, pooling of data is complicated by large heterogeneity in methods used across registries, definitions, and outcomes collected, which negatively impacts the ability to pool data [2]. The current study shows that if harmonization across registries in methods and collected outcomes can be reached, pooling of data will be possible for the majority of TK implants (93%), as only 7% of TK implants were used in a single registry, and this is particularly valuable for TK implants with limited sample size.
Combining data from multiple registries may also increase the heterogeneity of the included data due to factors other than recorded patient characteristics, where using data from a single registry may limit this heterogeneity, which makes interpretation more straightforward. For instance, revision tendencies can vary between countries, which influences the estimated performance (i.e., revision risks) of specific TK implants. When using data from an individual registry, such differences in tendencies to revise may be smaller, although between-hospital variations in revision thresholds may still exist as well as differences in operative volumes of individual surgeons and hospitals, all known as factors influencing revision risks [26,27]. Although data pooling has its limitations, we believe that pooling data should be recommended, to increase the number of implants for statistical power and thus to better represent real-world performance of a specific implant.
Limitations
First, we were limited in the patient characteristics that could be compared between registries, where more factors (e.g., American Society of Anesthesiologists [ASA] classification) may affect the safety and performance of primary TK implants, and are therefore important to take into account when comparing the performance of TK implants [28]. Second, the frequency of UK implants used in a registry may affect differences found in patient characteristics where it is known that UK implants are more commonly used, for instance in younger patients [29]. However, as the variation in UK implants used across registries was relatively small, the impact is likely negligible. Third, there could have been selection bias because not all TK implants used in patients were reported in registries (i.e., TK-implant-level completeness ranges from 58.7% to 97%). Lastly, we limited our analysis to 4 national registries where a larger number of regional, national, and multi-country registries exist [2]. Including additional registries could have resulted in a higher number of specific TK implants used across multiple registries for which patient characteristics could be compared.
Conclusion
Most TK implants were used in multiple registries, indicating that if harmonization of data collection across registries is achieved, this will enable pooling of data across registries for detection of safety concerns, particularly for those TK implants with limited sample size within a registry. In addition, differences in characteristics of patients receiving the same TK implant across registries were found, which should be considered when comparing the performance of the same TK implant across registries and may assist clinicians in implant selection for specific patients.
- de Steiger R N, Miller L N, Davidson D C, Ryan P, Graves S E. Joint registry approach for identification of outlier prostheses. Acta Orthop 2013; 84(4): 348-52. doi: 10.3109/17453674.2013.831320.
- Hoogervorst L A, Geurkink T H, Lübbeke A, Buccheri S, Schoones J W, Torre M, et al. Quality and uility of European cardiovascular and orthopaedic registries for the regulatory evaluation of medical device safety and performance across the implant lifecycle: a systematic review. Int J Health Policy Manag 2023; 12: 7648. doi: 10.34172/ijhpm.2023.7648.
- Von Hintze J, Ponkilainen V, W-Dahl A, Hailer N P, Furnes O, Fenstad A M, et al. Incidence of constrained condylar and hinged knee implants and mid- to long-term survivorship: a register-based study from the Nordic Arthroplasty Register Association (NARA). Acta Orthop 2025; 96: 142-50. doi: 10.2340/17453674.2025.42999.
- Hoogervorst L A, Ren Y, Melvin T, Stratton-Powell A A, Lübbeke A, Geertsma R E, et al. Safety notices and registry outlier data measure different aspects of safety and performance of total knee implants: a comparative study of safety notices and register outliers. Acta Orthop 2024; 95: 667-76. doi: 10.2340/17453674.2024.42361.
- Gausden E B, Puri S, Chiu Y F, Figgie M P, Sculco T P, Westrich G, et al. Mid-term survivorship of primary total knee arthroplasty with a specific implant. Bone Joint J 2023; 105-b(3): 277-83. doi: 10.1302/0301-620X.105B3.BJJ-2022-0616.R1.
- García-Crespo R, Marco F, Moro L E, Ariza G, Luque R, López-Durán L. Midterm results of Optetrak posterior-stabilized total knee system after 7 to 12 years in a university hospital. J Arthroplasty 2011; 26(8): 1326-31. doi: 10.1016/j.arth.2010.11.020.
- Puijk R, Sierevelt I N, Pijls B, Spekenbrink-Spooren A, Nolte P A. Increased risk of aseptic loosening for posterior stabilized compared with posterior cruciate-retaining uncemented total knee replacements: a cohort study of 13,667 knees from the Dutch Arthroplasty Registry. Acta Orthop 2023; 94: 600-6. doi: 10.2340/17453674.2023.33283.
- Schmerler J, Harris A B, Srikumaran U, Khanuja H S, Oni J K, Hegde V. Body mass index and revision total knee arthroplasty: does cause for revision vary by underweight or obese status? J Arthroplasty 2023; 38(12): 2504-9.e1. doi: 10.1016/j.arth.2023.06.019.
- Bayliss L E, Culliford D, Monk A P, Glyn-Jones S, Prieto-Alhambra D, Judge A, et al. The effect of patient age at intervention on risk of implant revision after total replacement of the hip or knee: a population-based cohort study. Lancet 2017; 389(10077): 1424-30. doi: 10.1016/S0140-6736(17)30059-4.
- Gademan M G J, Van Steenbergen L N, Cannegieter S C, Nelissen R, Marang-Van De Mheen P J. Population-based 10-year cumulative revision risks after hip and knee arthroplasty for osteoarthritis to inform patients in clinical practice: a competing risk analysis from the Dutch Arthroplasty Register. Acta Orthop 2021; 92(3): 280-4. doi: 10.1080/17453674.2021.1876998.
- Lingard E A, Katz J N, Wright E A, Sledge C B. Predicting the outcome of total knee arthroplasty. J Bone Joint Surg Am 2004; 86(10): 2179-86. doi: 10.2106/00004623-200410000-00008.
- Ackerman I N, Dieppe P A, March L M, Roos E M, Nilsdotter A K, Brown G C, et al. Variation in age and physical status prior to total knee and hip replacement surgery: a comparison of centers in Australia and Europe. Arthritis Rheum 2009; 61(2): 166-73. doi: 10.1002/art.24215.
- Irmola T, Ponkilainen V, Mäkelä K T, Robertsson O, A W D, Furnes O, et al. Impact of Nordic Arthroplasty Register Association (NARA) collaboration on demographics, methods and revision rates in knee arthroplasty: a register-based study from NARA 2000-2017. Acta Orthop 2022; 93:866-73. doi: 10.2340/17453674.2022.5256.
- Grimberg A W, Steinbrück A. [10 Years of the German Arthroplasty Registry-EPRD: what has been achieved?]. Orthopadie (Heidelb) 2023; 52(6): 463-71. doi: 10.1007/s00132-023-04385-3.
- Italian Arthroplasty Registry (RIAP). Annual Report 2021: English addendum. Available from: https://riap.iss.it/riap/en/activities/reports/2023/07/10/annual-report-2021-english-addendum/ (last date accessed: September 18, 2024).
- Dutch Arthroplasty Register (LROI). Online LROI annual report 2022. Available from: https://www.lroi.nl/media/3j2o5wjg/pdf-lroi-annual-report-2022.pdf (date last accessed: September 18, 2024).
- Greenland S, Senn S J, Rothman K J, Carlin J B, Poole C, Goodman S N, et al. Statistical tests, P values, confidence intervals, and power: a guide to misinterpretations. Eur J Epidemiol 2016; 31(4): 337-50. doi: 10.1007/s10654-016-0149-3.
- Pijls B G, Dekkers O M, Middeldorp S, Valstar E R, van der Heide H J, Van der Linden-Van der Zwaag H M, et al. AQUILA: assessment of quality in lower limb arthroplasty. An expert Delphi consensus for total knee and total hip arthroplasty. BMC Musculoskelet Disord 2011; 12: 173. doi: 10.1186/1471-2474-12-173.
- Hofstede S N, Gademan M G J, Stijnen T, Nelissen R, Marang-van de Mheen P J. The influence of preoperative determinants on quality of life, functioning and pain after total knee and hip replacement: a pooled analysis of Dutch cohorts. BMC Musculoskelet Disord 2018; 19(1): 68. doi: 10.1186/s12891-018-1991-0.
- Melvin T, Torre M. New medical device regulations: the regulator’s view. EFORT Open Rev 2019; 4(6): 351-6. doi: 10.1302/2058-5241.4.180061.
- Courage O, Strom L, van Rooij F, Lalevée M, Heuzé D, Papin P E, et al. Higher rates of surgical and medical complications and mortality following TKA in patients aged ≥ 80 years: a systematic review of comparative studies. EFORT Open Rev 2021; 6(11): 1052-62. doi: 10.1302/2058-5241.6.200150.
- Willigenburg N W, Poolman R W. The difference between statistical significance and clinical relevance: the case of minimal important change, non-inferiority trials, and smallest worthwhile effect. Injury 2023; 54(Suppl 5): 110764. doi: 10.1016/j.injury.2023.04.051.
- Kang Y, Liu C, Ji Y, Zhang H, Wang Y, Bi W, et al. The burden of knee osteoarthritis worldwide, regionally, and nationally from 1990 to 2019, along with an analysis of cross-national inequalities. Arch Orthop Trauma Surg 2024; 144(6): 2731-43. doi: 10.1007/s00402-024-05250-4.
- Günsche J L, Pilz V, Hanstein T, Skripitz R. The variation of arthroplasty procedures in the OECD countries: analysis of possible influencing factors by linear regression. Orthop Rev (Pavia) 2020; 12(3): 8526. doi: 10.4081/or.2020.8526.
- de Steiger R N, Hallstrom B R, Lübbeke A, Paxton E W, van Steenbergen L N, Wilkinson M. Identification of implant outliers in joint replacement registries. EFORT Open Rev 2023; 8(1): 11-17. doi: 10.1530/EOR-22-0058.
- Kugler C M, Goossen K, Rombey T, De Santis K K, Mathes T, Breuing J, et al. Hospital volume–outcome relationship in total knee arthroplasty: a systematic review and dose–response meta-analysis. Knee Surg Sports Traumatol Arthrosc 2022; 30(8): 2862-77. doi: 10.1007/s00167-021-06692-8.
- Pondugula P, Krumme J W, Seedat R, Patel N K, Golladay G J. Evaluation of painful total knee arthroplasty: an approach based on common etiologies for total knee arthroplasty revision. Musculoskelet Surg 2024; 108(1): 11-20. doi: 10.1007/s12306-023-00800-2.
- Stone B, Nugent M, Young S W, Frampton C, Hooper G J. The lifetime risk of revision following total knee arthroplasty: a New Zealand Joint Registry study. Bone Joint J 2022; 104-b(2): 235-41. doi: 10.1302/0301-620X.104B2.BJJ-2021-0890.R1.
- Hariri M, Hagemann M, Mick P, Deisenhofer J, Panzram B, Innmann M, et al. Physical activity of young patients following minimally invasive lateral unicompartmental knee replacement. J Clin Med 2023; 12(2). doi: 10.3390/jcm12020635.
- Dutch Arthroplasty Register (LROI). LROI Implant Library—Knee. Available from: https://www.lroi.nl/media/gzxorhtj/625522d75894blroi-implant-library-knee.pdf (date last accessed: September 18, 2024).