Precision of low-dose CT-based micromotion analysis technique for the assessment of early acetabular cup migration compared with gold standard RSA: a prospective study of 30 patients up to 1 year
Vasileios ANGELOMENOS 1,2, Maziar MOHADDES 1,2, Raed ITAYEM 1,2, and Bita SHAREGHI 1,2
1 Department of Orthopedics, Institute of Clinical Sciences, Sahlgrenska Academy, University of Gothenburg, Gothenburg; 2 Department of Orthopedics, Sahlgrenska University Hospital, Gothenburg, Sweden
Background and purpose — Computed tomography micromotion analysis (CTMA) can be used to determine implant micro-movements using low-dose CT scans. By using CTMA, a non-invasive measurement of joint implant movement is enabled. We evaluated the precision of CTMA in measuring early cup migration. Standard marker-based radiostereometric analysis (RSA) was used as reference. We hypothesised that CTMA can be used as an alternative to RSA in assessing implant micromotions.
Patients and methods — We included 30 patients undergoing total hip arthroplasty (THA). Acetabular cup migration at 1 year was measured with RSA and CTMA. To determine the precision of both methods, 20 double examinations (postoperatively) with repositioning of the patients were performed. The precision was calculated from zero by assuming that there was no motion of the prosthesis between the 2 examinations.
Results — The precision of RSA ranged from 0.06 to 0.15 mm for translations and 0.21° to 0.63° for rotations. Corresponding values for CTMA were 0.06 to 0.13 mm and 0.23° to 0.35°. A good level of agreement was found between the methods regarding cup migration and rotation at 1 year.
Interpretation — The precision of CTMA in measuring acetabular cup migration and rotation is comparable to marker-based RSA. CTMA could possibly thus be used as an alternative method to detect early implant migration.
Citation: Acta Orthopaedica 2022; 93: 459–465. DOI http://dx.doi.org/10.2340/17453674.2022.2528.
Copyright: © 2022 The Author(s). This is an Open Access article distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (https://creativecommons.org/licenses/by-nc/4.0/), allowing third parties to copy and redistribute the material in any medium or format and to remix, transform, and build upon the material for non-commercial purposes, provided proper attribution to the original work.
Submitted: 2021-11-27. Accepted: 2022-03-31. Published: 2022-04-22.
Correspondence: vasileios.angelomenos@vgregion.se
MMΟ conceptualized the study. All authors were involved in the study design. BSΗ retrieved and prepared the data. BSΗ and VAΝ performed statistical analyses. VAΝ wrote the manuscript, and all authors contributed with notable critical appraisal of the text and approved the final version.
Olof Sandberg, employee at SECTRA (Linköping, Sweden), provided technical help but had no part in collection and interpretation of the data.
Acta thanks Bart L Kaptein and Stephan Maximilian Röhrl for help with peer review of this study.
Early implant migration within the first 2 postoperative years after hip arthroplasty can be used as a predictor of future aseptic implant loosening (1-4). Marker-based radiostereometric analysis (RSA), the most accurate method for determining micromotions of orthopedic implants, has the disadvantage of requiring insertion of spherical tantalum markers into the bone or into the liner. The introduction, though, of model-based RSA solved the issue of prosthesis markers.
Computed tomography micromotion analysis (CTMA) (Sectra, Linköping, Sweden) is an analysing tool that can be used to determine implant micro-movements after implantation without the need for bone or implant markers. The principles of the method have been studied for more than 15 years (5-11). The technique is based on a simple principle, which is to obtain low-dose CT scans over time using thresholds to identify the bone and the implant, registering these objects and calculating migration. The CT scans are uploaded to a specially designed software that identifies movements and spatial and volumetric changes of 2 rigid bodies between 2 different CT examinations. Due to the lack of need for markers and reduced complexity of image acquisition, CTMA has been proposed as an alternative to marker-based RSA in measuring implant micro-movements after THA (5-7,9-11).
We evaluated the precision of measurements performed with CTMA compared with marker-based RSA and compared the reliability of the CTMA versus RSA in measuring cup migration within 1 year.
Patients and methods
30 patients (30 hips) with hip osteoarthritis scheduled for a cemented THA were included. The patients are part of a larger ongoing implant migration study at Sahlgrenska University Hospital, Mölndal, Sweden. The inclusion criteria in the current study were patients who had complete RSA examinations and CT scans on 2 occasions, postoperatively (double examinations) and at 1 year postoperatively. This patient cohort consisted of 14 males and 16 females with a mean age of 68 years (61–75). All patients received a cemented MS-30 (Zimmer GmbH, Rheinau, Germany) stem and a cemented Exceed (Biomet UK Ltd, Swindon, UK) cup. 2 different types of cement were used: Refobacin and Palacos.
5 patients could not be included in the 1 year follow-up: 1 patient was lost because of the Covid-19 pandemic (missing both RSA and CTMA), 1 patient was excluded due to low quality CT scans, and 3 patients due to very high condition number (CN) on the RSA analysis due to unsatisfactory marker placement; CTMA analysis was, however, possible for these 3 patients. Complete evaluation was performed for 26 patients with RSA and for 28 patients with CTMA at the 1-year follow-up.
Based on previous RSA research, we concluded that to measure precision 15–20 patients are sufficient (12). The precision of the methods was, thus, determined by documenting the differences between 20 double examinations postoperatively. Between the double RSA examinations, the patients were repositioned without moving the calibration cage and X-ray tubes (12). Correspondingly, between the double CTMA examinations, the patients were repositioned while the conditions of the CT apparatus remained unchanged. We assumed that no true migration of the prosthesis occurred between the 2 consecutive postoperative examinations.
Radiostereometric analysis (RSA)
RSA was used to evaluate the rotations and translations about the 3 orthogonal axes (x, y, z). The postoperative double examinations were performed within 3 days following surgery, with the patients in the supine position. Subsequent examinations were performed at 1 year after surgery. All examinations were done using an Adora radiographic system (NRT-Nordisk Røntgen Teknik A/S, Hasselager, Denmark) and the uniplanar technique with the RSA calibration cage under the examination table (cage 77, UmRSA Biomedical, Umeå, Sweden). Radiographic measurements and analysis were performed with UmRSA Digital Measure and UmRSA Analysis software version 7.0. All RSA measurements and analysis were conducted by a biomedicine scientist with long clinical and research experience. Complete evaluation of all radiographs was performed only if 3 or more tantalum markers of the segments corresponding to the acetabular bone and the cup could be identified with a scatter corresponding to a condition number (CN) less than 125 and a stability corresponding to a mean error of rigid body fitting (ME) of at most 0.35 mm (13,14).
Computed tomography micromotion analysis (CTMA)
Each patient was examined postoperatively (double examination) and at 1 year after the surgery. On each occasion, a double CT examination was performed, allowing movement of the lower body between the 2 exposures. All CT examinations were performed using a Discovery CT 750 HD scanner (GE Healthcare, Chicago, IL, USA). A defined CT protocol was applied with the following imaging parameters: 100 kV, tube current 15–100 mA (automatic), slice thickness 0.625 mm, increments 0.312 mm, pitch 0.984, rotation time 1 second, noise index 42.5, detector coverage 40 mm, reconstruction 0.625 mm. Evaluation of all CT scans was performed with analysis software (CTMA, Sectra, Linköping, Sweden) by a certified biomedicine scientist at our department. Prior to the analysis process, a protocol was defined regarding the measurement registration settings. No tantalum markers were used in any step of the CT analysis. Several scans of the included patients were randomly selected and observed to determine the most suitable settings for registration of the bone and the implant for this patient cohort. A threshold of 300 HU (Hounsfield) for bone and 1900 HU for the implant was set. The same thresholds were set for all double examinations. At 1 year, the threshold for the implant was adjusted for 5 patients (2000–2400 HU) and for 1 of these patients it was also adjusted for the bone registration (500 HU). The CT analysis process was done stepwise. First, 2 CT datasets of the same patient were uploaded into the CTMA software. The pelvic bone was registered as the reference body by setting the threshold to 300 HU and then a visual overlap of the pelvic bone was obtained (Figure 1A and 1B). The software matched the reference body in the 2 scans as closely as possible and provided a colour-coded overlay as visual assistance that helped the analyst to determine whether the matching process was performed properly or needs correction (Figure 1C). In the next step, the moving body, in this case the thread of the cup (that was used to represent the polyethylene cup) was registered by setting the threshold to 1900 HU. A visual overlap of the thread of the cup was then obtained (Figure 2A and 2B) and the position of the thread in the 2 scans was matched as closely as possible to each other (Figure 2C). Migration data of the movement was obtained in 6 degrees of freedom (translations along and rotations around X, Y, Z axes) (15).
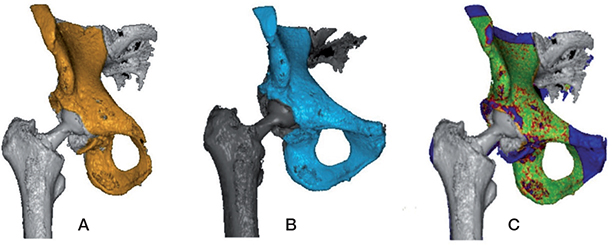
Figure 1. Registration of the pelvic bone as reference object on 2 different CT scans (A and B) and verification of the overlap process with the help of chromatic overlay (C).
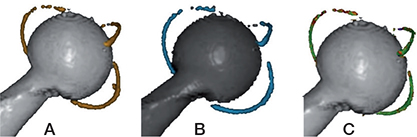
Figure 2. Registration of the acetabular cup ring as moving body on 2 different CT scans (A and B) and verification of the overlap process with the help of chromatic overlay (C).
Coordinate systems
The system of coordinates that is used in RSA and in CTMA is not identical. RSA makes use of an anatomical fixed coordinate system that is dependent on the calibration cage. CTMA uses a standard DICOM coordinate system that can be transformed to match that of RSA (Figure 3). To obtain a comparable coordinate system to RSA, the CT coordinate system was adjusted in a multi-planar reconstruction (MPR) view and the software then calculated the movement of the centre of mass (COM) of the metallic thread relative to the pelvic bone between the 2 examinations.
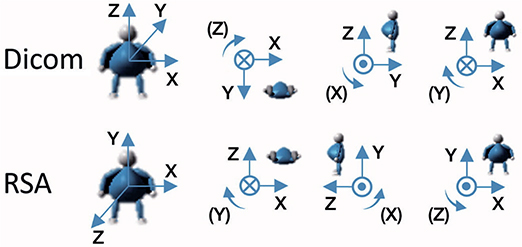
Figure 3. CT DICOM coordinate system of CTMA and coordinate system of RSA. The translations are positive in the direction of the arrow, and the rotations are positive in a clockwise direction.
Radiation dose
The mean effective radiation dose for the scans used in the CTMA analysis was estimated to be 0.8 mSv (0.5–1.2 mSv) per scan. The corresponding mean effective dose of RSA was estimated to be 0.2 mSv per scan.
Statistics
The statistical analysis was performed using IBM SPSS version 27.0.0 software (IBM Corp, Armonk, NY, USA). Descriptive statistics was used to describe the outcome variables at the measurements point. The precision of the measurements was defined as the degree to which repeated measurements under unchanged conditions show the same results and it refers to random errors only (16,17). The precision was measured using the standard deviation (SD) of the differences calculated between double examinations multiplied by the critical value (t) obtained from the T-table adjusted for the number of observations minus 1 (n–1) (Precision = SD x t(n–1)) (18). The precision was calculated by assuming that there was no motion of the implant between the examinations (18,19). The total migration was calculated by using the Pythagorean theorem (total translation = √(X2+Y2+Z2)) (20). The total rotation was calculated using the simplified Euler’s rotation theorem (total rotation = √(θX 2+θY 2+θΖ 2)), which can be used when micromotions are studied (21,22) . Given the small sample size in this study, normality of the differences between the methods was tested using a graphical investigation. We evaluated the distribution of the differences for each paired comparison with a histogram with a density curve. All paired differences were judged to be roughly normally distributed. The median and range were calculated at 1 year for migration measured with RSA and CTMA. All tests used were 2-sided. To assess the agreement between marker-based RSA and CTMA at 1 year follow-up, Bland–Altman plot analysis was used. The plots describe the average between the same measurements on the x-axis in relation to the differences between the same measurements on the y-axis. In the Bland–Altman analysis, the bias and limits of agreement, including the respective 95% confidence intervals, are reported. Our primary outcome measure was set as the proximal/distal cup migration, which translates to migrations along the y-axis (Figure 3).
Ethics, funding, data sharing, and potential conflict of interest
All patients included gave written informed consent to participate in the study. Approval for the study was obtained through the Regional Ethics Review Board in Gothenburg, entry number 801-17. Sharing of data is available upon request, though personal information of patients cannot be disclosed. No funding was received for this study. No conflict of interest has occurred.
Results
Clinical precision (double examinations)
The precision for the RSA measurements of cup migration ranged between 0.06 and 0.15 mm for translations and between 0.21º and 0.63º for rotations. Corresponding values for CTMA measurements ranged between 0.06 and 0.13 mm and between 0.23º and 0.35º. The precision for the total migration was 0.18mm for RSA and 0.16 mm for CTMA (Table 1). The precision for the total rotation was 0.46 º for RSA and 0.38 º for CTMA.
Cup migration at 1-year follow-up
The median (range) translations in the y-axis at 1 year measured with RSA were 0.13 mm (–0.10–1.75). Corresponding values for measurements performed with CTMA were 0.11 mm (–0.15–2.00). The median (range) total migration was 0.21 mm (0.05–2.50) for RSA and 0.22 mm (0.40–2.23) (Table 2). At 1 year, the median cup rotations were small (–0.11˚ to 0.06˚). Detailed values for cup translation and rotation at 1 year for RSA and CTMA on the 3 orthogonal axes x, y, and z are described on Table 2. The 95% limits of agreement (LoA) on the y-axis were –0.22 mm (lower) and 0.25 mm (upper). Between these LoA, roughly 95% of all paired measurements on the y-axis appear to be included (23-27). The Bland–Altman plots at 1 year (Figure 4 and 5) showed on the orthogonal axes that the measured discrepancy between the methods and specifically the measurements for translations on the y-axis (which is this study’s primary outcome measure) are within the limits of clinical importance (0.2–1 mm for proximal/distal translations) (2,3) and margin of error for RSA. On the rest of the axes, the measured discrepancy was roughly under or within the limits of clinical importance for translation and margin of error for RSA and under or within the precision values for rotation for both methods (Table 1). According to the Bland–Altman plots presented in the current study, the best agreement between the methods was observed in the proximal/distal (y-axis) and total migration (3D) measurements.
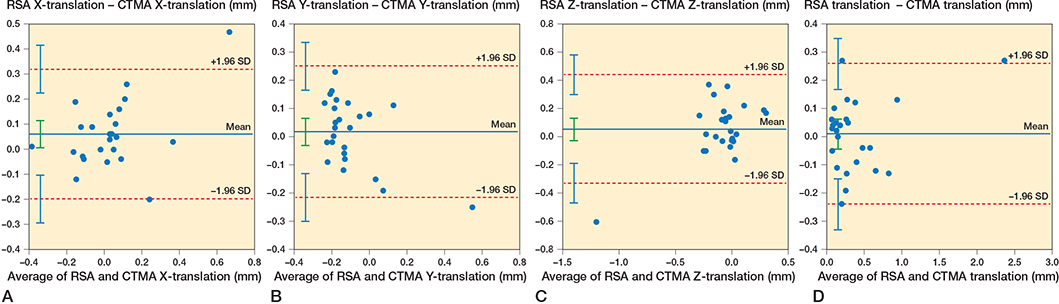
Figure 4. Bland–Altman plot for translations on the X-axis (A), Y-axis (B), Z-axis (C), and total translation along the 3 orthogonal axes (D). Limits of agreement are shown as dotted red lines with 95% confidence intervals in light blue. Bias is shown as a solid blue line with a 95% confidence interval in green.
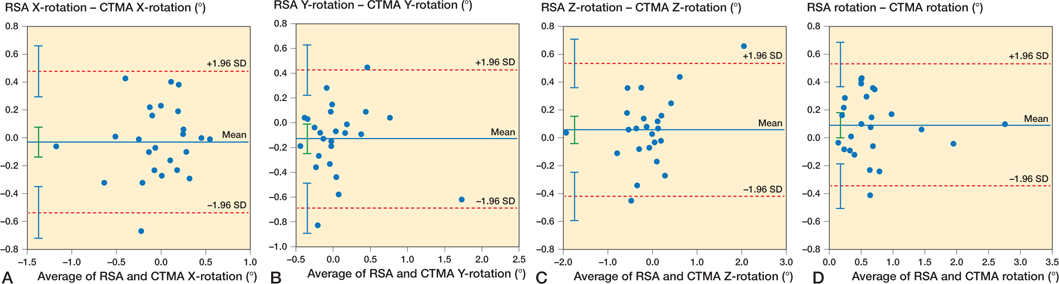
Figure 5. Bland–Altman plot for rotation on the X-axis (A), Y-axis (B), Z-axis (C), and total rotation around the 3 orthogonal axes (D). Limits of agreement are shown as dotted red lines with 95% confidence intervals in light blue. Bias is shown as a solid blue line with a 95% confidence interval in green.
Discussion
This is the first study evaluating the precision and migration pattern of acetabular cups using RSA and CTMA in the same cohort of patients. The majority of previous RSA implant studies of the hip that refer to a limit of clinical importance of 0.2–1 mm regarding this as a predictor of future aseptic implant loosening refer to proximal/distal translations along the y-axis (2,3,28,29). This is important to underline even in this study, as our primary outcome measure was translations along the y-axis. Our findings suggest that the precision of CTMA in measuring proximal cup migration postoperatively (double examinations) and up to 1 year is comparable to the standard RSA and can be considered for assessment of early implant migration. This finding is in accordance with previous experimental and clinical studies (6,30).
Brodén et al. (31) measured the precision of the CTMA method between 0.07 and 0.31 mm for translations and 0.20º and 0.39º for rotations. In the current study, CTMA displayed a slightly higher precision for both translations and rotations.
RSA enables 3-D measurements of implant micro-movement with respect to the host bone that might be undetectable on plain radiographs. Several studies have shown that proximal cup migration exceeding 0.2–1 mm or more within 2 years postoperatively measured with RSA can be used to predict the risk of implant loosening (2,3,28,29). Due to its high accuracy and the ability to measure the 3-D position of an object, RSA has been acknowledged as a gold standard method for assessing micro-movements of orthopedic implants in vivo. However, the disadvantages of marker-based RSA, such as the necessity to use spherical tantalum markers attached to the implant or inserted into the bone, tantalum markers being over-projected by the implant (32), or the need for specially designed radiographic equipment have raised interest in alternative methods that can overcome the drawbacks of the RSA method without sacrificing the precision and accuracy of the analysis and measurements. A partial solution to some of these problems was the introduction of model-based RSA, where a 3-D surface contour of the true implant is being used instead of markers to determine migration and wear (33). However, this method requires information on the 3-D surface of each individual size used in the study and the accuracy of the poseestimation algorithm depends on both implant design and the accuracy of surface model that is used (34).
CT, as a 3-D imaging modality, has the advantage of elimination of superimposition of objects outside the area of interest, high image resolution, and the ability to discern different tissues with ease. The high resolution came at the beginning though at a cost: higher radiation dose. Through the years, advancements such as low-dose protocols and metal artifact reduction (MAR) protocols have turned the tables (35,36), making the use of CT more widespread. CT-based micromotion analysis has been studied for the past 15 years (5-11) and is reported to be an accurate analyzing tool for the evaluation of implant micro-movements after THA, and is also considered as a potential alternative to the gold standard RSA.
Experimental studies using phantoms, in vivo studies with pilot patients (5,6,37-40) and clinical studies (30,31,40) over the last few years have shown that, with methods using low-dose CT scans, precision can be achieved that is very much comparable to RSA while radiation dosage also stays at low levels (41). In previous studies the mean effective dose for CTMA has been reported at 0.33 mSv for an experimental hip study (6) and 0.2–2.3 mSv for a clinical hip study (31). The mean effective radiation dose in the current study was 0.8 mSv (0.5–1.2 mSv) for CTMA, in accordance with the European guidelines on exposure in medical and biomedical research, “Radiation protection 99” (European Commission, 1998) (42). In the current study radiation doses within these limits were achieved. Even though the effective radiation dose for a CT examination is higher than that for RSA, we reckon that the advantages of the CT-based method outweigh this fact. According to our data, CTMA could be considered as an alternative to RSA in studying proximal cup migration, although, in order to achieve acceptable radiation dose for patients in a long-term follow-up migration study, further optimization of CT-scan protocols and adjustment of radiation dose is necessary to reduce the radiation exposure (43).
An advantage of the CTMA method is that there is no need for tantalum markers, as bone or implant surfaces are used in the analysis instead. Thus, considerations regarding marker over-projection and patient exclusion due to few markers or high condition number are overcome. Using low-dose CT scans for assessment of implant micro-movements and CTMA as an analyzing tool facilitates measuring implant migration. These advantages could also mean that it would be possible to include patients even retrospectively after the implantation is performed.
Another advantage of CTMA is that the equipment needed, namely a CT apparatus, is available even in smaller institutes. Furthermore, CTMA, using a predetermined scan protocol reduces the need for an experienced radiographer and the presence of trained personnel.
A drawback of CTMA is the lack of the condition number and mean error as in RSA (13,14). With CTMA, the correctness of the analysis and measurements is determined by the color-coding system provided by the software (30). In that essence, the assessment is based solely on the analyst’s or user’s experience. However, based on previous research even a relatively inexperienced CTMA user can produce excellent results with very high inter- and intra-observer repeatability (15). Nonetheless, we highly recommend that training in CTMA is completed beforehand.
In this study we did not confirm inter-observer repeatability, because the same analyst performed all the RSA and CTMA measurements. We suggest that in future studies inter-observer repeatability should also be validated.
In this study cemented acetabular cups were examined. That said, it is difficult to directly extrapolate the results of this study to other joints. Parameters such as metal artefacts, radiation doses, and prosthesis design, among many, should be taken into account.
In conclusion, the precision of the CTMA method, according to our data, is as trustworthy as that of RSA.
- Pijls B G, Valstar E R, Nouta K A, Plevier J W M, Fiocco M, Middeldorp S, et al. Early migration of tibial components is associated with late revision. Acta Orthop 2012; 83(6): 614-24. doi: 10.3109/17453674.2012.747052.
- Klerken T, Mohaddes M, Nemes S, Kärrholm J. High early migration of the revised acetabular component is a predictor of late cup loosening: 312 cup revisions followed with radiostereometric analysis for 2–20 years. Hip Int 2015; 25(5): 471-6. doi: 10.5301/hipint.5000246.
- Pijls B G, Nieuwenhuijse M J, Fiocco M, Plevier J W M, Middeldorp S, Nelissen R G H H, et al. Early proximal migration of cups is associated with late revision in THA: a systematic review and meta-analysis of 26 RSA studies and 49 survival studies. Acta Orthop 2012; 83(6): 583-91. doi: 10.3109/17453674.2012.745353.
- van der Voort P, Pijls B G, Nieuwenhuijse M J, Jasper J, Fiocco M, Plevier J W M, et al. Early subsidence of shape-closed hip arthroplasty stems is associated with late revision. Acta Orthop 2015; 86(5): 575-85. doi: 10.3109/17453674.2015.1043832.
- Olivecrona H, Maguire GQ, Noz M E, Zeleznik M P, Kesteris U, Weidenhielm L. A CT method for following patients with both prosthetic replacement and implanted tantalum beads: preliminary analysis with a pelvic model and in seven patients. J Orthop Surg Res 2016; 11(1): 27. doi: 10.1186/s13018-016-0360-7.
- Brodén C, Olivecrona H, Maguire G Q, Noz M E, Zeleznik M P, Sköldenberg O. Accuracy and precision of three-dimensional low dose CT compared to standard RSA in acetabular cups: an experimental study. BioMed Res Int 2016; 2016: 5909741. doi: 10.1155/2016/5909741.
- Otten V, Maguire G Q, Noz M E, Zeleznik M P, Nilsson K G, Olivecrona H. Are CT scans a satisfactory substitute for the follow-up of RSA migration studies of uncemented cups? A comparison of RSA double examinations and CT datasets of 46 total hip arthroplasties. BioMed Res Int 2017; 2017. doi: 10.1155/2017/3681458.
- Bakhshayesh P, Olivecrona H, Weidenhielm L, Enocson A. A new CT based method for post-operative motion analysis of pelvic fractures. 3D Research 2019; 10(1): 1-9. doi: 10.1007/s13319-019-0217-z.
- Olivecrona H, Olivecrona L, Weidenhielm L, Noz M E, Hansen J K, Maguire G Q, et al. A new technique for diagnosis of acetabular cup loosening using computed tomography: preliminary experience in 10 patients. Acta Orthop 2008; 79(3): 346-53. doi: 10.1080/17453670710015247.
- Olivecrona H, Olivecrona L, Weidenhielm L, Noz M E, Maguire G Q, Zeleznik M P, et al. Stability of acetabular axis after total hip arthroplasty, repeatability using CT and a semiautomated program for volume fusion. Acta Radiol 2003; 44(6): 653-61. doi: 10.1046/j.1600-0455.2003.00153.x.
- Olivecrona L, Olivecrona H, Weidenhielm L, Noz M E, Maguire G Q, Zeleznik M P. Model studies on acetabular component migration in total hip arthroplasty using CT and a semiautomated program for volume merging. Acta Radiol 2003; 44(4): 419-29. doi: 10.1034/j.1600-0455.2003.00086.x.
- Valstar E R, Gill R, Ryd L, Flivik G, Börlin N, Kärrholm J. Guidelines for standardization of radiostereometry (RSA) of implants. Acta Orthop 2005; 76(4): 563-72. doi: 10.1080/17453670510041574.
- Söderkvist I, Wedin P Å. Determining the movements of the skeleton using well-configured markers. J Biomech 1993; 26(12): 1473-7. doi: 10.1016/0021-9290(93)90098-Y.
- Söderkvist I, Wedin P Å. On condition numbers and algorithms for determining a rigid body movement. BIT 1994; 34(3): 424-36. doi:10.1007/BF01935651.
- Sandberg O, Tholén S, Carlsson S, Wretenberg P. The anatomical SP-CL stem demonstrates a non-progressing migration pattern in the first year: a low dose CT-based migration study in 20 patients. Acta Orthop 2020; 91(6): 654-9. doi: 10.1080/17453674.2020.1832294/SUPPL_FILE/IORT_A_1832294_SM5111.MP4.
- Sköldenberg O, Eisler T, Stark A, Muren O, Martinez-Carranza N, Ryd L. Measurement of the migration of a focal knee resurfacing implant with radiostereometry. Acta Orthop 2014; 85(1): 79-83. doi: 10.3109/17453674.2013.869654.
- Ranstam J. Methodological note: Accuracy, precision, and validity. Acta Radiol 2008; 49(1): 105-6. doi: 10.1080/02841850701772706.
- Nebergall A K, Rader K, Palm H, Malchau H, Greene M E. Precision of radiostereometric analysis (RSA) of acetabular cup stability and polyethylene wear improved by adding tantalum beads to the liner. Acta Orthop 2015; 86(5): 563-8. doi: 10.3109/17453674.2015.1054255.
- Börlin N, Röhrl S M, Bragdon C R. RSA wear measurements with or without markers in total hip arthroplasty. J Biomech 2006; 39(9). doi: 10.1016/j.jbiomech.2005.05.004.
- Derbyshire B, Prescott R J, Porter M L. Notes on the use and interpretation of radiostereometric analysis. Acta Orthop 2009; 80(1): 124-30. doi: 10.1080/17453670902807474.
- Selvik G. Roentgen stereophotogrammetry: a method for the study of the kinematics of the skeletal system. Acta Orthop Scand 1989; 60(S232): 1-51. doi: 10.3109/17453678909154184.
- Millán Vaquero R M, Vais A, Lynch S D, Rzepecki J, Friese K I, Hurschler C, et al. Helical axis data visualization and analysis of the knee joint articulation. J Biomech Eng 2016; 138(9). doi: 10.1115/1.4034005/371279.
- Giavarina D. Understanding Bland Altman analysis. Biochem Medica 2015; 25(2): 141-51. doi: 10.11613/BM.2015.015.
- Myles P S, Cui J. I. Using the Bland–Altman method to measure agreement with repeated measures. Brit J Anaesth 2007; 99(3): 309-11. doi: 10.1093/bja/aem214.
- Bland J M, Altman D G. Comparing methods of measurement: why plotting difference against standard method is misleading. Lancet 1995; 346(8982): 1085-7. doi: 10.1016/S0140-6736(95)91748-9.
- Bland J M, Altman D G. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet 1986; 327(8476). doi: 10.1016/S0140-6736(86)90837-8.
- Bland J M, Altman D G. Measuring agreement in method comparison studies. Stat Methods Med Res 1999; 8(2): 135-60. doi: 10.1177/096228029900800204.
- Kärrholm J, Borssen B, Löwenhielm G, Snorrason F. Does early micromotion of femoral stem prostheses matter? 4–7-year stereoradiographic follow-up of 84 cemented prostheses. J Bone Joint Surg Br 1994; 76-B(6). doi: 10.1302/0301-620X.76B6.7983118.
- Kärrholm J. Radiostereometric analysis of early implant migration: a valuable tool to ensure proper introduction of new implants. Acta Orthop 2012; 83(6): 551-2. doi: 10.3109/17453674.2012.745352.
- Brodén C, Sandberg O, Olivecrona H, Emery R, Sköldenberg O. Precision of CT-based micromotion analysis is comparable to radiostereometry for early migration measurements in cemented acetabular cups. Acta Orthop 2021; 92(4): 419-23. doi: 10.1080/17453674.2021.1906082/SUPPL_FILE/IORT_A_1906082_SM2295.PDF.
- Brodén C, Sandberg O, Sköldenberg O, Stigbrand H, Hänni M, Giles J W, et al. Low-dose CT-based implant motion analysis is a precise tool for early migration measurements of hip cups: a clinical study of 24 patients. Acta Orthop 2020; 91(3): 260-5. doi: 10.1080/17453674.2020.1725345.
- Kaptein B L, Valstar E R, Stoel B C, Rozing P M, Reiber J H C. A new type of model-based Roentgen stereophotogrammetric analysis for solving the occluded marker problem. J Biomech 2005; 38(11): 2330-4. doi: 10.1016/j.jbiomech.2004.09.018.
- Valstar E R, de Jong F W, Vrooman H A, Rozing P M, Reiber J H C. Model-based Roentgen stereophotogrammetry of orthopaedic implants. J Biomech 2001; 34(6): 715-22. doi:10.1016/S0021-9290(01)00028-8.
- Seehaus F, Emmerich J, Kaptein B L, Windhagen H, Hurschler C. Experimental analysis of model-based roentgen stereophotogrammetric analysis (MBRSA) on four typical prosthesis components. J Biomech Eng 2009; 131(4): 041004. doi: 10.1115/1.3072892/459921.
- Trattner S, Pearson G D N, Chin C, Cody D D, Gupta R, Hess C P, et al. Standardization and optimization of CT protocols to achieve low dose. J Am Coll Radiol 2014; 11(3): 271-8. doi: 10.1016/j.jacr.2013.10.016.
- Gjesteby L, de Man B, Jin Y, Paganetti H, Verburg J, Giantsoudi D, et al. Metal artifact teduction in CT: where are we after four decades? IEEE Access 2016; 4. doi: 10.1109/ACCESS.2016.2608621.
- Scheerlinck T, Polfliet M, Deklerck R, van Gompel G, Buls N, Vandemeulebroucke J. Development and validation of an automated and marker-free CT-based spatial analysis method (CTSA) for assessment of femoral hip implant migration: in vitro accuracy and precision comparable to that of radiostereometric analysis (RSA). Acta Orthop 2016; 87(2): 139-45. doi: 10.3109/17453674.2015.1123569.
- Eriksson T, Maguire G Q, Noz M E, Zeleznik M P, Olivecrona H, Shalabi A, et al. Are low-dose CT scans a satisfactory substitute for stereoradiographs for migration studies? A preclinical test of low-dose CT scanning protocols and their application in a pilot patient. Acta Radiol 2019; 60(12): 1643-52. doi: 10.1177/0284185119844166.
- Brodén C, Giles J W, Popat R, Fetherston S, Olivecrona H, Sandberg O, et al. Accuracy and precision of a CT method for assessing migration in shoulder arthroplasty: an experimental study. Acta Radiol 2020; 61(6): 776-82. doi: 10.1177/0284185119882659.
- Goldvasser D, Hansen V J, Noz M E, Maguire G Q, Zeleznik M P, Olivecrona H, et al. In vivo and ex vivo measurement of polyethylene wear in total hip arthroplasty. Acta Orthop 2014; 85(3): 271-5. doi: 10.3109/17453674.2014.913225.
- Sandgren B, Skorpil M, Nowik P, Olivecrona H, Crafoord J, Weidenhielm L, et al. Assessment of wear and periacetabular osteolysis using dual energy computed tomography on a pig cadaver to identify the lowest acceptable radiation dose. Bone Joint Res 2016; 5(7): 307-13. doi: 10.1302/2046-3758.57.2000566.
- Radiation protection 99 (European Commission, 1998) https://ec.europa.eu/energy/sites/ener/files/documents/099_en.pdf
- Blom I F, Koster L A, Brinke B ten, Mathijssen N M C. Effective radiation dose in radiostereometric analysis of the hip. Acta Orthop 2020; 91(4): 390-395. doi: 10.1080/17453674.2020.1767443/SUPPL_FILE/IORT_A_1767443_SM7929.PDF.