Risk for re-revision and type of antibiotic-loaded bone cement in hip or knee arthroplasty revisions: report of the Dutch Arthroplasty Register
Pieter K BOS 1, Anneke SPEKENBRINK-SPOOREN 2, Peter CROUGHS 3, Sita M A BIERMA-ZEINSTRA 1,4, Max REIJMAN 1, and Jakob VAN OLDENRIJK 1
1 Department of Orthopedics and Sports Medicine, Erasmus MC University Medical Center, Rotterdam; 2 Dutch Arthroplasty Register (Landelijke Registratie Orthopedische Interventies), ’s-Hertogenbosch; 3 Department of Medical Microbiology and Infectious Diseases, Erasmus MC University Medical Center, Rotterdam; 4 Department of General Practice, Erasmus MC University Center Rotterdam, Rotterdam, the Netherlands
Background and purpose — High-dose dual antibiotic-loaded bone cement (ALBC) may reduce the risk of revision after total hip and knee replacements. The aim of our study therefore was to determine the risk of re-revision following first time aseptic hip or knee revision using single versus dual ALBC.
Patients and methods — Patients from the Dutch Arthroplasty Register treated from 2007 to 2018 with first time cemented aseptic hip (n = 2,529) or knee revisions (n = 7,124) were incorporated into 2 datasets. The primary endpoint of this observational cohort study was subsequent all-cause re-revision. Multivariable Cox proportional hazard and competing risk was analyzed for both groups.
Results — There was no difference in re-revision rate (any reason) with single versus dual ALBC (hazard ratio 1.06, 95% confidence interval [CI] 0.83–1.35 for hip and 0.93, CI 0.80–1.07 for knee revisions). The 10-year crude cumulative re-revision rate also showed no differences for single versus dual ALBC use. The crude cumulative 7-year THA re-revision and 9-year TKA re-revision rates did not show any difference in implant survival for common cement types used.
Conclusion — We could not confirm the potential benefit of using dual ALBC compared with single ALBC for aseptic hip and knee revisions.
Citation: Acta Orthopaedica 2023; 94: 471–476. DOI: https://doi.org/10.2340/17453674.2023.18645.
Copyright: © 2023 The Author(s). This is an Open Access article distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (https://creativecommons.org/licenses/by-nc/4.0/), allowing third parties to copy and redistribute the material in any medium or format and to remix, transform, and build upon the material for non-commercial purposes, provided proper attribution to the original work.
Submitted: 2023-01-01. Accepted: 2023-08-17. Published: 2023-09-20.
Correspondence: .k.bos@erasmusmc.nl
PKB, JvO, MR, PC, and SMAB wrote the study protocol and analysis plan. AS constructed 2 datasets from LROI and advised on survival analysis. PKB wrote the first draft of the manuscript, and all authors and collaborators revised the draft and approved the final manuscript.
The authors would like to thank Dr Sara Baart, Department of Biostatistics, for statistical advice regarding survival analysis.
Handling co-editors: Keijo Mäkelä and Robin Christensen
Acta thanks Heather A Prentice and Michael Charles Wyat for help with peer review of this manuscript.
Adapting treatment protocols, with a change from low-dose single antibiotic-loaded bone cement (ALBC) into high-dose dual ALBC could significantly reduce infection risk in hip and knee revision arthroplasty patients. Current evidence for the use of dual ALBC for arthroplasty patients consists of a single randomized clinical trial concerning hip fracture hemiarthroplasty patients, which shows a significant reduction in the 1-year infection rate when dual ALBC (gentamicin and clindamycin) was used instead of single ALBC (gentamicin) [1]. For aseptic revision arthroplasty, the evidence is limited to 2 low-level-evidence reports: one uncontrolled pilot cohort study [2], and one retrospective comparative study [3], both suggesting an improved outcome with dual ALBC use. The current data on the usage and effectiveness of dual ALBC for revision hip and knee surgery is thus limited.
The aim of our study was to determine the risk for re-revision following first time aseptic hip or knee revisions, using data from the Dutch Arthroplasty Registry (LROI). Furthermore, we aim to analyze the risk for re-revision based on the specific cement type used.
Patients and methods
The Dutch Arthroplasty Register (LROI) has 100% hospital coverage, with completeness of revision hip and knee arthroplasty of ≥ 97% (since 2015) [4]. Implant survival is determined based on registration of revision procedures and the survival of patients obtained from Vektis, the Dutch insurance healthcare database [5].
In the LROI, implant and bone cement characteristics are linked from the implant library to all procedures in the LROI based on registered article numbers [6]. This study is reported according to the STROBE guidelines for observational studies [7].
Patients, cement, and outcomes
2 datasets were composed from the LROI containing all patients with cemented hip or knee revisions, for all reasons excluding infection. Revision arthroplasties for infection were not included because the selection of antibiotics in these patients is likely targeted on the determined microorganisms and not used for prophylaxis. Patients with registered article numbers for bone cement were included in our dataset analyses. Orthopedic reconstructive studies generally require a minimum follow-up period of 2 years. We therefore included all registered revision hip and knee arthroplasties between the beginning of the LROI in 2007 and 2018 for outcome analysis. This results in a study timeframe ranging from January 1, 2007 until January 1, 2020.
We were interested in the association of ALBC choice and subsequent incidence of re-revision in first time hip and knee revisions. To exclude possible previous revisions for unknown causes we selected first revision arthroplasties for which the index procedure was registered in the LROI. Revision procedures were defined as operations in which 1 or more components of the primary prosthesis are exchanged.
The primary endpoint was re-revision for any reason in hip or knee revisions during the follow-up period in relation to the use of single ALBC versus dual ALBC, with a minimum follow-up of 2 years. Secondary to this, we analyzed re-revision for infection and the risk for re-revision based on the specific composition of the ALBC.
Bone cement
The current most frequently registered bone cement types were calculated from LROI annual reported use of separately packed and pre-packed bone cements [5]. In the Netherlands, during the 10-year follow-up period the proportion of cemented hip revisions was 55% and cemented knee revisions 85% [5]. To determine patterns in ALBC use across operative years the relative use of single versus dual ALBC per year was calculated for both datasets.
Statistics
Baseline data was calculated for each of the 2 databases using SPSS version 25 (IBM Corp, Armonk, NY, USA, 2017). Survival of hip and knee revisions was calculated as the time between first revision and first re-revision arthroplasty for any reason and first re-revision for infection for the period of the study (2007–2020). The falsifiable hypothesis is proportion implant survival with single ALBC = proportion implant survival with dual ALBC. The proportional hazards assumption for regression analysis was checked and met by inspecting Kaplan–Meier (KM) curves. Multivariable Cox proportional hazard analyses were performed to compare re-revision rates between patients treated with single and dual ALBC. Adjustments were made for sex, age, and ASA classification. Results were reported as unadjusted and adjusted hazard ratios (HR) with 95% confidence intervals (CI).
Cumulative crude incidence of re-revision was calculated using competing risk analysis, where death was considered to be a competing risk [8], with 95% confidence intervals (CI). The period for survival analysis for which more than around 50 patients are at risk in each group was 7 years for THA and 9 years for TKA patients.
P < 0.05 were considered statistically significant.
Ethics, funding, and disclosures
The research protocol was approved by the scientific committee of the LROI and is following the regulations of the LROI. All data is anonymous, therefore ethical approval from our local ethical commission was not applicable. We did not receive any funding for this study. JvO has an advisory consulting agreement with Heraeus, not related to the subject of this paper. Complete disclosure of interests statement forms according to ICMJE are available at the article page, doi: 10.2340/17453674.2023.18645
Results
Patient and bone cement characteristics
Between January 1, 2007 and January 1, 2018, 2,529 primary hip arthroplasties and 7,124 primary knee arthroplasties were followed by a first revision (Figure 1 and Table 1).
| Variables | Aseptic revision THA (n = 2,529) | Aseptic revision TKA (n = 7,124) | ||
| Single ABLC | Dual ABLC | Single ABLC | Dual ABLC | |
| Revisions | 1,669 (66) | 860 (34) | 5,270 (74) | 1,854 (26) |
| Male sex | 508 (30) | 282 (33) | 1,620 (31) | 579 (31) |
| BMI a, mean (SD) | 27.0 (4.7) | 27.0 (4.8) | 30.4 (5.2) | 30.5 (5.5) |
| Age, mean (SD) | 71.2 (11.0) | 69.5 (12.0) | 66.3 (9.4) | 65.2 (9.5) |
| Smoking a | 114 (6.8) | 66 (7.7) | 380 (7.2) | 137 (7.4) |
| Age groups | ||||
| < 50 | 68 (4.1) | 54 (6.3) | 206 (3.9) | 83 (4.5) |
| 50–59 | 160 (9.6) | 109 (13) | 1,045 (20) | 434 (23) |
| 60–69 | 456 (27) | 224 (26) | 1,996 (38) | 707 (38) |
| 70–79 | 593 (36) | 301 (35) | 1,589 (30) | 520 (28) |
| > 80 | 392 (26) | 170 (20) | 427 (8.1) | 109 (5.9) |
| Missing data | 2 (0.08) | 8 (0.1) | ||
| ASA classification | ||||
| I | 227 (14) | 140 (16) | 608 (12) | 245 (13) |
| II | 942 (56) | 473 (55) | 2,987 (57) | 1,053 (57) |
| III–IV | 453 (27) | 229 (27) | 683 (13) | 241 (13) |
| Missing data | 65 (2.6) | 1,307 (18) | ||
| BMI classification a | ||||
| < 18.5 | 17 (1.0) | 13 (1.5) | 13 (0.2) | 3 (0.2) |
| Normal, 18.5–25 | 328 (20) | 178 (21) | 484 (9.2) | 165 (8.9) |
| Overweight, 25–30 | 382 (23) | 222 (26) | 1243 (24) | 470 (25) |
| Obese, 30–40 | 195 (12) | 114 (13) | 1,539 (29) | 502 (27) |
| > 40 | 12 (0.7) | 7 (0.8) | 142 (2.7) | 74 (4.0) |
| Missing data | 1,061 (42) | 2,370 (33) | ||
| a Recorded since 2014 Single ABLC = single antibiotic loaded cement, Dual ABLC = dual antibiotic loaded cement. Numbers do not add up to 100% due to missing data. |
||||
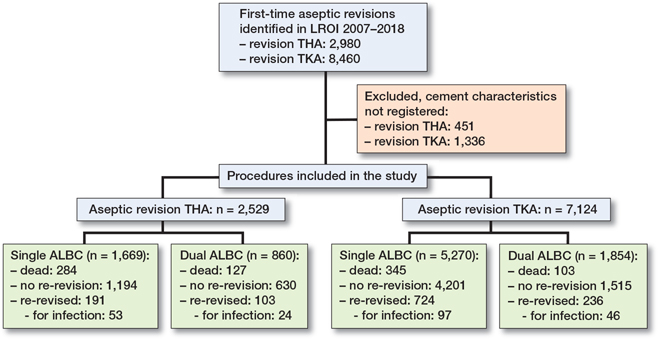
Figure 1. Flowchart of study. THA = total hip arthroplasty; TKA = total knee arthroplasty.
Cement compound was not registered in 14.7% for revision THA and 14.6% for revision TKA patients. Dual ALBC was used in 860 patients (34%) for hip revisions and 1,854 patients (26%) for knee revisions (Table 2). The bone cement characteristics and percentage of ALBC types used during the study period (2007–2018) are given in Table 3. The current most frequently registered bone cement types published in the annual LROI report are given in Table 4 (see Appendix) [5]. We did not see large variation across years for relative use of single versus dual ALBC (Figure 2, see Appendix).
| Factor | Aseptic revision THA | Aseptic revision TKA | ||
| Single ABLC | Dual ABLC | Single ABLC | Dual ABLC | |
| Revisions, n | 1,669 | 860 | 5,270 | 1,854 |
| Re-revision for | ||||
| any reason, n (%) | 191 (11) | 103 (12) | 724 (14) | 236 (13) |
| Crude HR (CI) | 1 | 1.06 (0.83–1.35) | 1 | 0.93 (0.80–1.07) |
| Adjusted a HR (CI) | 1 | 1.01 (0.79–1.29) | 1 | 1.13 (0.97–1.30) |
| Re-revision for | ||||
| infection, n (%) | 53 (3.2) | 24 (2.8) | 97 (1.8) | 46 (2.5) |
| Crude HR (CI) | 1 | 0.86 (0.55–1.43) | 1 | 1.35 (0.95–1.91) |
| Adjusted a HR (CI) | 1 | 0.86 (0.53–1.40) | 1 | 1.31 (0.93–1.87) |
| a Adjustment for age, sex, and ASA classification registered during entire inclusion period. For Abbreviations, see Table 1. |
||||
Re-revision rates
The percentage of re-revision for any reason with use of single versus dual ALBC in the revision THA dataset was 11% and 12% (HR dual ALBC 1.06 (CI 0.83–1.35) and in the revision TKA dataset 14% and 13% (HR dual ALBC 0.93 (CI 0.80–1.07). The majority of re-revisions are due to causes other than infection (Table 2). There is no statistically significant difference in re-revision rate with dual versus single ALBC use.
Survival analysis
Single ALBC versus dual ALBC
The 10-year crude cumulative first re-revision rate for hip and knee revisions showed no difference between single and dual ALBC use (Table 2 and Figures 3a and b).
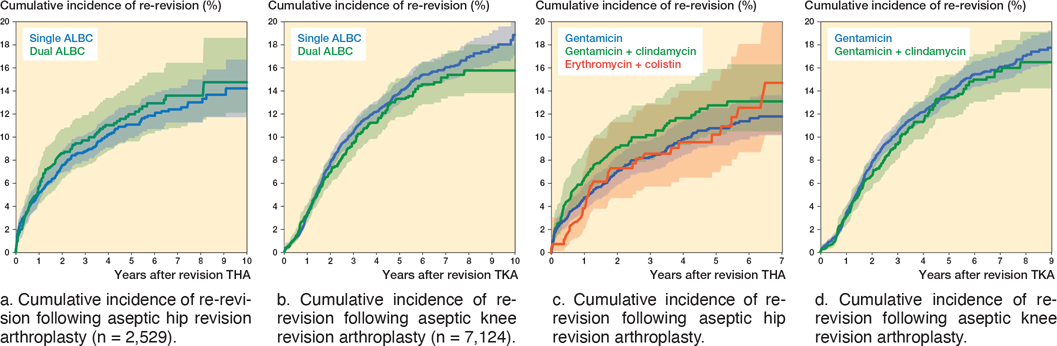
Figure 3. Cumulative incidence of re-revision following aseptic revision arthroplasty with use of single or dual ABLC and with most frequently used bone cement types in the Netherlands in 2007–2018. Shaded area represent the 95% confidence interval.
In the revision THA dataset, the most frequently used bone cement types were gentamicin, gentamicin + clindamycin, and erythromycin + colistin, whereas in the revision TKA dataset gentamicin and gentamicin + clindamycin were used most frequently (Table 3).
Hip revision arthroplasty
The crude cumulative 7-year re-revision rate showed no differences between any of the cement types. The crude cumulative 7-year re-revision rate in revision THA using gentamicin ALBC was 12% (CI 10–14, n = 354 patients at risk at 7 years), using gentamicin + clindamycin ALBC 13% (CI 11–16, n = 102 patients at risk at 7 years), and using erythromycin + colistin ALBC 15% (CI 10–21, n = 49 patients at risk at 7 years) (Figure 3c).
Knee revision arthroplasty
The crude cumulative 9-year re-revision rate showed no differences between any of the cement types. The crude cumulative 9-year re-revision rate in revision TKA using gentamicin ALBC was 18% (CI 16–19, n = 327 patients at risk at 9 years), and using gentamicin + clindamycin ALBC 17% (CI 14–19, n = 67 patients at risk at 9 years) (Figure 3d).
Discussion
The aim of the study was to determine the risk for re-revision following first time aseptic hip or knee revisions and single versus dual ALBC, using Dutch Arthroplasty Registry data. We showed no association between increased revision implant survival and the use of dual ALBC versus single ALBC bone cement, with a minimum of 2-year follow-up. Adjusting for age, sex, and ASA classification did not change our results. With the small CI in both groups, it is unlikely that the lack of difference between groups is due to a “Type 2 error.” We found comparable re-revision rates up to 7- and 9-years’ follow-up for hip and knee revision patients following first revision with most frequently used bone cement types.
The major advantage of this registry study is that we can verify the possible improvement of care (introduction of high-dose ALBC) with available data within a national joint registry. Antibiotic loading in bone cement has been part of treatment protocols in many European countries for decades [9,10], whereas in the USA cost-effectiveness studies did not convince the FDA to allow its use for low-risk primary arthroplasty due to high costs [9,11]. A recent literature review with meta-analysis demonstrated a reduced risk for development of PJI when using ALBC versus plain bone cement in THA [12]. A lower all-cause revision rate and revision for PJI rate was also shown at 5-year follow-up with ALBC use in a large retrospective case control study, comparing 4,741 TKA implanted with plain bone cement versus 11,231 TKA implanted with ALBC [13]. Another retrospective study, however, could not detect a reduced PJI rate when comparing 1,434 TKA implanted with plain bone cement with 1,077 TKA implanted with ALBC [14]. This will be further elucidated in a large prospective register-based RCT aimed to study the value of routine ALBC use in TKA [11]. In a recent analysis in the Kaiser Joint Replacement Registry (87,018 TKA replacements) the additional cost of ALBC was not justified but the risk of PJI in diabetic patients was reduced [15].
Changing prophylactic ALBC, based on initial study results [1,2], by adding antibiotics and increasing the doses used should be introduced carefully considering the current evidence and debate on antibiotic loading of bone cement. Patient selection, regional antibiotic resistance differences, and national cost differences appear to be important factors for treatment choice. Data from national registries and accumulating data from cohort studies and RCTs will take us further in our ambition to provide the best long-term care for arthroplasty patients.
In general, for implant studies, 2-year follow up is required. A minimum follow-up of 2 years was chosen for all patients analyzed in this LROI registry study. With these 2 datasets we were able to reliably compare the outcome of implant survival in relation to the use of gentamicin single ALBC, gentamicin + clindamycin dual ALBC, and gentamicin + colistin dual ALBC (THA only). The use of gentamicin + vancomycin ALBC has been limited in the Netherlands in the previous 10 years, which makes reliable estimation of the effect of adding vancomycin to bone cement not possible in the current datasets.
Limitations
Patients who underwent revision for infection were excluded from our analysis because the selection of antibiotics in these patients is likely targeted on the determined microorganisms. Preoperatively grown microorganisms are not registered in the LROI database.
Information on individual infection risk or infections in medical history and vulnerability of patients may be overrepresented in the dual ALBC use groups. Also, information on perioperative, systemic antibiotic prophylaxis, which is routinely given and standard care in treatment protocols, is not registered in LROI. However, the relatively large number of subjects analyzed in this study may give a better estimation of the effect of dual ALBC in general compared with the presented limited-size case series.
Development of antibiotic resistance probably plays an important role in the long-term outcome of arthroplasty patients. A disadvantage of our registry study is that information on microorganisms and antibiotic sensitivity is not available. Patients with a PJI in the hemiarthroplasty study showed an increased proportion of resistance to ciprofloxacin and clindamycin in the dual ALBC group, whereas due to the significantly lower PJI rate in the dual ALBC group the absolute resistance rate was lower [16].
Not only is the selection and concentration of antibiotics in bone cement important, but also other characteristics may affect antibiotic release and cement quality and thereby influence risk for revision [17]. The current study provides insights into the use of antibiotic-loaded cement in the Netherlands and its possible influence on re-revision rates. Comparing international registry data may reveal variation in eradication success rates based on differences in antibiotic loading of cement and national microbiologic profiles [18].
Changing prophylaxis in revision hip and knee arthroplasty protocols, at higher costs and with possible side effects such as increased antibiotic resistance development, should be scientifically substantiated.
Conclusion
We could not show a difference in all-cause re-revision and re-revision for infection rates for revision hip or knee arthroplasty patients with the use of dual ALBC compared with single ALBC, with a minimum of 2-year follow-up. With 7- and 9-years’ follow-up for hip and knee revision patients we also found comparable re-revision rates for the individual bone cement types. The results of this study do not confirm the observed benefit of dual ALBC use in revision cases.
- Sprowson A P, Jensen C, Chambers S, Parsons N R, Aradhyula N M, Carluke I, et al. The use of high-dose dual-impregnated antibiotic-laden cement with hemiarthroplasty for the treatment of a fracture of the hip: the Fractured Hip Infection trial. Bone Joint J 2016; 98-B(11): 1534-41. doi: 10.1302/0301-620X.98B11.34693.
- Abdelaziz H, von Forster G, Kuhn K D, Gehrke T, Citak M. Minimum 5 years’ follow-up after gentamicin- and clindamycin-loaded PMMA cement in total joint arthroplasty. J Med Microbiol 2019; 68(3): 475-9. doi: 10.1099/jmm.0.000895.
- Sanz-Ruiz P, Matas-Diez J A, Villanueva-Martinez M, Santos-Vaquinha Blanco A D, Vaquero J. Is dual antibiotic-loaded bone cement more effective and cost-efficient than a single antibiotic-loaded bone cement to reduce the risk of prosthetic joint infection in aseptic revision knee arthroplasty? J Arthroplasty 2020; 35(12): 3724-9. doi: 10.1016/j.arth.2020.06.045.
- LROI. Data quality completeness. Annual report; 2020.
- LROI. Dutch Arthroplasty Registry. Online LROI annual report; 2020.
- van Steenbergen L N, Denissen G A, Spooren A, van Rooden S M, van Oosterhout F J, Morrenhof J W, et al. More than 95% completeness of reported procedures in the population-based Dutch Arthroplasty Register. Acta Orthop 2015; 86(4): 498-505. doi: 10.3109/17453674.2015.1028307.
- von Elm E, Altman D G, Egger M, Pocock S J, Gotzsche P C, Vandenbroucke J P, et al. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. Int J Surg 2014; 12(12): 1495-9. doi: S1743-9191(14)00212-X [pii]10.1016/j.ijsu.2014.07.013.
- Lacny S, Wilson T, Clement F, Roberts D J, Faris P D, Ghali W A, et al. Kaplan–Meier survival analysis overestimates the risk of revision arthroplasty: a meta-analysis. Clin Orthop Relat Res 2015; 473(11): 3431-42. doi: 10.1007/s11999-015-4235-8 10.1007/s11999-015-4235-8 [pii].
- Hansen E N, Adeli B, Kenyon R, Parvizi J. Routine use of antibiotic laden bone cement for primary total knee arthroplasty: impact on infecting microbial patterns and resistance profiles. J Arthroplasty 2014; 29(6): 1123-7. doi: 10.1016/j.arth.2013.12.004.
- Engesaeter L B, Lie S A, Espehaug B, Furnes O, Vollset S E, Havelin L I. Antibiotic prophylaxis in total hip arthroplasty: effects of antibiotic prophylaxis systemically and in bone cement on the revision rate of 22,170 primary hip replacements followed 0-14 years in the Norwegian Arthroplasty Register. Acta Orthop Scand 2003; 74(6): 644-51. doi: 10.1080/00016470310018135.
- Leta T H, Gjertsen J E, Dale H, Hallan G, Lygre S H L, Fenstad A M, et al. Antibiotic-loaded bone cement in prevention of periprosthetic joint infections in primary total knee arthroplasty: a register-based multicentre randomised controlled non-inferiority trial (ALBA trial). BMJ Open 2021; 11(1): e041096. doi: 10.1136/bmjopen-2020-041096.
- Farhan-Alanie M M, Burnand H G, Whitehouse M R. The effect of antibiotic-loaded bone cement on risk of revision following hip and knee arthroplasty. Bone Joint J 2021; 103-B(1): 7-15. doi: 10.1302/0301-620X.103B1.BJJ-2020-0391.R1.
- Bendich I, Zhang N, Barry J J, Ward D T, Whooley M A, Kuo A C. antibiotic-laden bone cement use and revision risk after primary total knee arthroplasty in U.S. veterans. J Bone Joint Surg Am 2020; 102(22): 1939-47. doi: 10.2106/JBJS.20.00102.
- Yayac M, Rondon A J, Tan T L, Levy H, Parvizi J, Courtney P M. The economics of antibiotic cement in total knee arthroplasty: added cost with no reduction in infection rates. J Arthroplasty 2019; 34(9): 2096-101. doi: 10.1016/j.arth.2019.04.043.
- Namba R S, Prentice H A, Paxton E W, Hinman A D, Kelly M P. Commercially prepared antibiotic-loaded bone cement and infection risk following cemented primary total knee arthroplasty. J Bone Joint Surg Am 2020; 102(22): 1930-8. doi: 10.2106/JBJS.19.01440.
- Tyas B, Marsh M, Oswald T, Refaie R, Molyneux C, Reed M. Antibiotic resistance profiles of deep surgical site infections in hip hemiarthroplasty: comparing low dose single antibiotic versus high dose dual antibiotic impregnated cement. J Bone Jt Infect 2018; 3(3): 123-9. doi: 10.7150/jbji.22192jbjiv03p0123 [pii].
- Monzon R A, Coury J G, Disse G D, Lum Z C. Bone cement in total hip and knee arthroplasty. JBJS Rev 2019; 7(12): e6. doi: 10.2106/JBJS.RVW.19.00031.
- Antimicrobial Resistance C. Global burden of bacterial antimicrobial resistance in 2019: a systematic analysis. Lancet 2022; 399(10325): 629-55. doi: 10.1016/S0140-6736(21)02724-0.
Supplementary data
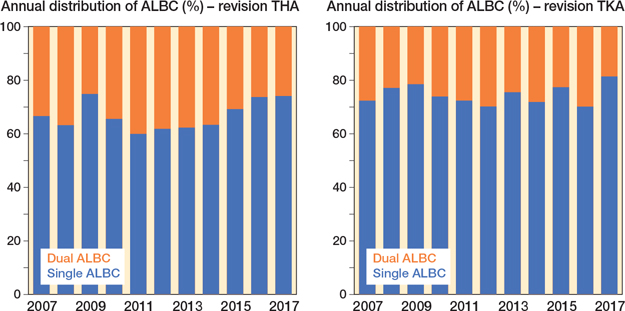
Figure 2. Proportion (%) of bone cement type used per year for revision hip or knee arthroplasty.