The obesity paradox and mortality after pathological hip fractures: a Swedish registry study
Jessica EHNE 1, Panagiotis TSAGOZIS 1,2, Anja LIND 3, Rikard WEDIN 1,2, and Margareta HEDSTRÖM 1,3
1 Department of Trauma and Reparative Medicine, Karolinska University Hospital, Stockholm; 2 Department of Molecular Medicine and Surgery, Karolinska Institutet, Stockholm; 3 Department of Clinical Science, Intervention and Technology, Karolinska Institutet, Stockholm, Sweden
Background and purpose — Obesity as measured by BMI has been associated with increased survival in various diseases, a phenomenon known as the “obesity paradox.” It is unknown whether obesity is associated with survival after pathological fractures. We investigated the association between BMI and survival after surgery for pathological hip fracture, to improve survival prognostication, and lay grounds for further interventional nutritional studies.
Patients and methods — We analyzed prospectively collected data from Swedish nationwide registry “RIKSHÖFT.” The study cohort included 1,000 patients operated for a pathological hip fracture between 2014 and 2019. BMI registered on admission was available in 449 patients. Overall patient survival was measured according to the Kaplan– Meier method. Multivariable regression was used to evaluate association with other potential factors that influence patient survival.
Results — Overweight and obesity were associated with an increased postoperative survival in male patients with surgically treated pathological hip fractures. Multivariable analysis considering potential confounders confirmed this finding. The association was not that strong in women and did not reach statistical significance.
Interpretation — BMI, a commonly available clinical parameter, is a good predictor of overall survival for patients operated on for pathological hip fracture. Incorporation of BMI in existent survival prognostication algorithms should be considered. Treatment of malnutrition in this frail group of patients is worth studying.
Citation: Acta Orthopaedica 2022; 93: 185–189. DOI http://dx.doi.org/10.2340/17453674.2021.1020.
Copyright: © 2021 The Author(s). This is an Open Access article distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (https://creativecommons.org/licenses/by-nc/4.0/), allowing third parties to copy and redistribute the material in any medium or format and to remix, transform, and build upon the material for non-commercial purposes, provided proper attribution to the original work.
Submitted: 2021-01-14. Accepted: 2021-10-17. Published: 2022-01-03.
Correspondence: jessica.ehne@sll.se
PT, AL, and JE analyzed the data and wrote the manuscript. RW and MH had the role of supervisors and have given continuous feedback during the process. MH also conceived and planned the research.
Acta thanks Marie Anneberg Brahefor help with peer review of this study.
Metastatic bone disease is common among cancer patients. Approximately 10% of them suffer a pathological fracture and approximately one-third of them die within a year after surgery (1,2). Treatment of pathological hip fractures is surgical. The choice of implant and adjuvant postoperative treatment depends on the estimated postoperative patient survival (3).
Although pre-obesity and obesity are associated with a higher risk of death in the general population, a reverse phenomenon called the “obesity paradox” has been observed in some but not all types of cancer (4,5). There are studies which suggest that the obesity paradox is also seen in traumatic fractures and hip replacement surgery (6–11).
Various patient survival prognostication models have been developed to aid the clinician with preoperative planning. The most used include factors such as patient age, the extent of metastatic spread, type of primary malignancy and laboratory values such as blood hemoglobin levels, but none includes BMI (12–14).
Considering the ambiguity regarding the obesity paradox in cancer and the findings in traumatic fracture surgery, we set out to explore whether there was an association between BMI and postoperative survival in a nationwide cohort of patients operated on for pathological fractures of the hip. The hypothesis was that overweight or obesity would be associated with increased postoperative survival.
Patients and methods
We conducted a registry-based nationwide cohort study on patients operated on for a pathological hip fracture due to metastasis between January 1, 2014, and December 31, 2018. Patient data was extracted from the Swedish National Registry for Hip Fractures (RIKSHÖFT) (Figure 1). The study is reported according to STROBE guidelines.

Figure 1. Study cohort and patient inclusion ending with final study population, n = 1,000; BMI was available in n = 449 of these. Patients included from Swedish national registry RIKSHÖFT.
Reporting to RIKSHÖFT is optional and takes place at 47 of the 52 orthopedic clinics in Sweden. Coverage rate in comparison with the Swedish National Patient Register exceeded 80% during the years 2014–2018 (National Board of Health and Welfare). Data collection at baseline is done by nurses or secretaries and a follow-up questionnaire is administered 4 months after index surgery.
The following variables are collected upon hospital arrival: age, sex, fracture date, fracture type, surgical method, walking ability, walking aids, cognitive function, and residency. Comorbidity is assessed using the ASA score as part of standard preoperative practice (15,16).
BMI registration is optional and calculated based on the patient’s weight and height measured on admission to hospital. BMI was further divided categorically according to the World Health Organization (WHO) guidelines into underweight ≤ 18.4, normal weight 18.5–24.9, overweight 25.0–29.9, and obesity ≥ 30.0. Information on date of death has been linked to RIKSHÖFT from Statistics Sweden using the unique personal identification number assigned to all individuals living in Sweden.
Subgroup analysis
High ASA and male sex have been associated with increased mortality in studies of non-pathological hip fractures and we therefore analyzed them separately (17–20). Age was divided into 2 groups, above and below the median value. ASA grading was formed into 2 groups, ASA1–2 and ASA 3–4. ASA 5 was excluded due to only 1% of patients belonging to this subgroup.
Statistics
We analyzed categorical data with the chi-square test. Survival analysis was done as per Kaplan and Meier, and we used the logrank test to evaluate differences between groups. Correlation between the survival time and BMI was also done in a Cox regression model with hazard ratios (HRs) and 95% confidence intervals (CI) estimated for each BMI group in relation to the normal weight group. We adjusted analysis for ASA score, sex, and age. A p-value < 0.05 was considered as statistically significant. Statistical calculations were performed in SPSS 25 (IBM Corp, Armonk, NY, USA).
Ethics, data sharing, funding, and potential conflicts of interests
The study was approved by the regional Ethics Committee of Stockholm Dnr 2017/1088-31. Primary data can be shared upon reasonable request by MH. This research has received funding from Stockholm County ALF fund. The authors have no conflicts of interest to declare.
Results
Cohort
We included 1,000 patients (Table 1). BMI on hospital admission was documented in 449 patients (underweight 35, normal weight 224, overweight 135, and obese 55) (Figure 2). There was no statistically significant correlation between BMI and sex (p = 0.9), age (p = 0.1), treatment method (p = 0.5), time to surgery (p = 1), or type of fracture (p = 0.8). BMI was significantly higher (p = 0.03) in patients living independently than for patients admitted from nursing homes or already hospitalized (Figure 2).

Figure 2. BMI distribution of 449 patients operated on for a pathological fracture of the hip (left panel). Boxplot of BMI distribution among patients operated on for a pathological fracture of the hip, depending on the admission status of each patient. Patients coming from nursing homes or hospitals (n = 129) had a statistically significantly lower BMI than those being admitted from home (n = 320) (right panel).
Correlation of BMI with postoperative mortality
Median overall patient survival time was 10 months (CI 8–12) and mean overall survival 23 months (CI 21–25). In the Kaplan–Meier survival analysis, patients with overweight and obesity had longer postoperative survival (p = 0.02) (Figure 3). Other potential prognostic factors such as age, sex, and ASA score were also evaluated regarding their association with patient survival (Table 2). In a multivariable model including these factors only BMI retained its prognostic significance (Table 3).
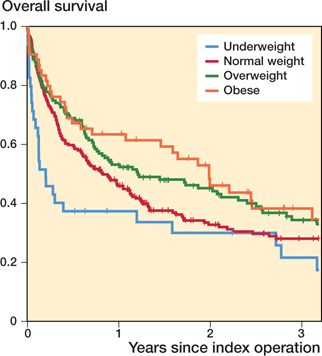
Figure 3. Kaplan–Meier survival analysis. Patients with overweight and obesity had increased survival compared with both patients of normal weight and underweight patients.
The probabilities for survival for males differed between BMI groups. Women had slightly increased (not statistically significant) postoperative survival with increasing BMI (Figure 4).

Figure 4. Kaplan–Meier survival analysis. The effect of BMI on survival reached statistical significance in men (left panel) but not in women (right panel).
Since BMI is only optionally recorded in the Swedish National Registry for Hip Fractures, and information was missing in 449/1,000 patients, we conducted a sensitivity analysis to assess reporting bias. We found no statistically significant difference in the distribution of BMI between hospitals that had an almost complete registration of BMI (≥ 75%) and the rest of the reporting units (p = 0.6).
Discussion
The major finding of our study is that the obesity paradox holds true in patients treated for pathological hip fractures. This could be attributed to the extra metabolic reserve of overweight and obese patients, who apparently have not entered the catabolic state of disseminated cancer and thus have better survival chances.
Strengths of this study are its sample size and the quality of the registered data regarding BMI and ASA, as well as complete dataset on mortality. As RIKSHOFT is a nationwide registry, included patients were treated at all hospital levels, minimizing the risk of selection bias. The major limitation is that half of patients lacked information on BMI. Our sensitivity analyses as well as an earlier report from the same database have not shown any selection bias behind the missing information on BMI, and we regard this as an issue attributed to administrative reasons (10). Another limitation is that the cancer diagnosis and dissemination status of each patient was not recorded. On the other hand, some of the potential methodological limitations that have been proposed to explain the obesity paradox in previous studies such as inconsistencies in time of BMI assessment or the presence of inhomogeneous populations regarding race/ethnicity were not present in ours (5).
The obesity paradox is quite consistently described in traumatic fractures, although its interpretation is a matter of debate (10). Whether obesity confers a survival benefit in cancer patients is controversial and the pathophysiological mechanisms are not known. Theoretically, excess adipose tissue may provide for a metabolic reserve to be used in stress conditions, either chronic ones such as cancer or acute ones such as a fracture of a long bone and its surgical treatment. However, it cannot be excluded that being overweight may simply indicate the absence of illness-related weight loss (5). This raises concern that the “obesity paradox” itself may simply be attributed to confounding factors and biased statistical methods because the extensive research in this area suffers from confounding issues, which are difficult to adjust for (21). In our study, we adopted a stepwise analysis and tried to compensate for possible confounding factors that have been described in the relevant medical literature, such as comorbidity grade (ASA), age, and sex. Other factors that have been proposed as confounders of the obesity paradox are smoking habits, which are not recorded in the registry and thus could not be included, and the presence of malignancy, which was a prerequisite to be included in the study.
In our study the obesity paradox was obvious in the male population, while in women the effect did not reach statistical significance. This is in line with the findings of a previous study in which women with breast cancer who suffered fragility fractures were studied (22). It is possible that our study is underpowered regarding the female subgroup, yet another explanation could be that in breast cancer, the leading cause of cancer-related death in females, a high BMI is associated with a negative effect on survival, and this might eradicate the postulated protective factor of having extra energy reserves to cope with the acute catabolic stress of trauma and orthopedic surgery (22,23). This, however, remains to be shown in a larger cohort or in cohorts where the type of malignancy is known.
It is not possible to draw any firm conclusions on the nature of the obesity paradox itself from our study. Our results are, however, of clinical importance as they support the hypothesis that preoperative BMI could be integrated in existing models for estimation of postoperative survival. Importantly, the data call for clinical studies focusing on the treatment of malnutrition in these patients, and the effect of this intervention on survival.
Conclusion
BMI can be used for estimation of survival after surgically treated pathological fractures; inclusion in survival prognostication tools may increase their accuracy.
- Roodman G D. Mechanisms of bone metastasis. N Engl J Med 2004; 350(16): 1655-64. doi: 10.1056/nejmra030831
- Hansen B H, Keller J, Laitinen M, Berg P, Skjeldal S, Trovik C et al. The Scandinavian Sarcoma Group Skeletal Metastasis Register: survival after surgery for bone metastases in the pelvis and extremities. Acta Orthop Scand 2004; 75(311, Suppl.): 11-15. doi: 10.1080/00016470410001708270
- Bauer H C. Controversies in the surgical management of skeletal metastases. J Bone Joint Surg Br 2005; 87(5): 608-17. doi: 10.1302/0301620x.87b5.16021
- Lennon H, Sperrin M, Badrick E, Renehan A G. The obesity paradox in cancer: a review. Curr Oncol Rep 2016; 18(9): 56. doi: 10.1007/s11912-016-0539-4
- Park Y, Peterson L L, Colditz G A. The plausibility of obesity paradox in cancer: point. Cancer Res 2018; 78(8): 1898-1903. doi: 10.1158/00085472.can-17-3043.
- Prieto-Alhambra D, Premaor M O, Avilés F F, Castro A S, Javaid M K, Nogués X et al. Relationship between mortality and BMI after fracture: a population-based study of men and women aged ≥ 40 years. J Bone Miner Res 2014; 29(8): 1737-44 . doi: 10.1002/jbmr.2209
- Flodin L, Laurin A, Lökk J, Cederholm T, Hedström M. Increased 1-year survival and discharge to independent living in overweight hip fracture patients: a prospective study of 843 patients. Acta Orthop 2016; 87(2): 146-51. doi: 10.3109/17453674.2015.1125282
- Shaparin N, Widyn J, Nair S, Kho I, Geller D, Delphin E. Does the obesity paradox apply to early postoperative complications after hip surgery? A retrospective chart review. J Clin Anesth 2016; 32: 84-91. doi: 10.1016/j.jclinane.2015.12.037
- Akinleye S D, Garofolo G, Culbertson M D, Homel P, Erez O. The role of BMI in hip fracture surgery. Geriatr Orthop Surg Rehabil 2018; 129: 2151458517747414. doi: 10.1177/2151458517747414
- Modig K, Erdefelt A, Mellner C, Cederholm T, Talbäck M, Hedström M. “Obesity Paradox” holds true for patients with hip fracture: a registrybased cohort study. J Bone Joint Surg Am 2019; 101(10): 888-95. doi: 10.2106/jbjs.18.01249
- Bzovsky S, Comeau-Gauthier M, Schemitsch E H, Swiontkowski M, Heels-Ansdell D, Frihagen F, et al. Factors associated with mortality after surgical management of femoral neck fractures. J Orthop Trauma 2020; 34 Suppl 3:S15-S21. doi: 10.1097/bot.0000000000001937
- Bauer H C, Wedin R. Survival after surgery for spinal and extremity metastases. Prognostication in 241 patients. Acta Orthop Scand 1995; 66(2): 143-6. doi: 10.3109/17453679508995508
- Katagiri H, Takahashi M, Wakai K, Sugiura H, Kataoka T, Nakanishi K. Prognostic factors and a scoring system for patients with skeletal metastasis. J Bone Joint Surg Br 2005; 87(5): 698-703. doi: 10.1302/0301620x.87b5.15185
- Forsberg J A, Eberhardt J, Boland P J, Wedin R, Healey J H. Estimating survival in patients with operable skeletal metastases: an application of a Bayesian belief network. PLoS One 2011; 6(5): e19956. doi: 10.1371/journal.pone.0019956
- Dripps R D, Lamont A, Eckenhoff J E. The role of anesthesia in surgical mortality. JAMA 1961; 178: 261-6. doi: 10.1001/jama.1961.03040420001001
- Daabiss M. American Society of Anaesthesiologists physical status classification. Indian J Anaesth 2011; 55(2): 111-15. doi: 10.4103/0019-5049.79879
- Wehren L E, Hawkes W G, Orwig D L, Hebel J R, Zimmerman S I, Magaziner J. Gender differences in mortality after hip fracture: the role of infection. J Bone Miner Res 2003; 18(12): 2231-7 . doi: 10.1359/jbmr.2003.18.12.2231
- Endo Y, Aharonoff G B, Zuckerman J D, Egol K A, Koval K J. Gender differences in patients with hip fracture: a greater risk of morbidity and mortality in men. J Orthop Trauma 2005; 19(1): 29-35. doi: 10.1097/00005131-200501000-0000
- Johansen A, Tsang C, Boulton C, Wakeman R, Moppett I. Understanding mortality rates after hip fracture repair using ASA physical status in the National Hip Fracture Database. Anaesthesia 2017; 72(8): 961-6 . doi: 10.1111/anae.13908
- Liu Y, Peng M, Lin L, Liu X, Qin Y, Hou X. Relationship between American Society of Anesthesiologists (ASA) grade and 1-year mortality in nonagenarians undergoing hip fracture surgery. Osteoporos Int 2015; 26(3): 1029-33. doi: 10.1007/s00198-014-2931-y
- Banack H R, Stokes A. The “obesity paradox” may not be a paradox at all. Int J Obes (Lond) 2017; 41(8): 1162-3 . doi: 10.1038/ijo.2017.99
- Chan D S M, Vieira A R, Aune D, Bandera E V, Greenwood D C, McTiernan A, et al. Body mass index and survival in women with breast cancer: systematic literature review and meta-analysis of 82 follow-up studies. Ann Oncol 2014; 25(10): 1901-14 . doi: 10.1093/annonc/mdu042
- Blair C K, Wiggins C L, Nibbe A M, Storlie C B, Prossnitz E R, Royce M, et al. Obesity and survival among a cohort of breast cancer patients is partially mediated by tumor characteristics. NPJ Breast Cancer 2019; 5: 33. doi: 10.1038/s41523-019-0128-4